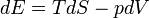Laws of thermodynamics
From Wikipedia, the free encyclopedia
The laws of thermodynamics, in principle, describe the specifics for the transport of heat and work in thermodynamic processes. Since their inception, however, these laws have become some of the most important in all of physics and other branches of science connected to thermodynamics.
There are generally considered to be four laws:
- The zeroth law of thermodynamics, which underlies the definition of temperature.
- The first law of thermodynamics, which mandates conservation of energy, and states in particular that heat is a form of energy.
- The second law of thermodynamics, which states that the entropy of the universe always increases, or (equivalently) that perpetual motion machines are impossible.
- The third law of thermodynamics, which concerns the entropy of an object at absolute zero temperature, and implies that it is impossible to cool a system all the way to exactly absolute zero.
Over the last 80 years or so, occasionally, various writers have suggested additional Laws, but none of them have been widely accepted.
Contents |
[edit] Zeroth law
If two thermodynamic systems are each in thermal equilibrium with a third, then they are in thermal equilibrium with each other.
When two systems are put in contact with each other, there will be a net exchange of energy between them unless or until they are in thermal equilibrium, that is, they are at the same temperature. While this is a fundamental concept of thermodynamics, the need to state it explicitly as a law was not perceived until the first third of the 20th century, long after the first three laws were already widely in use, hence the zero numbering. The Zeroth Law asserts that thermal equilibrium, viewed as a binary relation, is an equivalence relation.
[edit] First law
Energy can neither be created nor destroyed. It can only change forms.
In any process, the total energy of the universe remains the same.
For a thermodynamic cycle the net heat supplied to the system equals the net work done by the system.
The First Law states that energy cannot be created or destroyed; rather, the amount of energy lost in a steady state process cannot be greater than the amount of energy gained. This is the statement of conservation of energy for a thermodynamic system. It refers to the two ways that a closed system transfers energy to and from its surroundings - by the process of heating (or cooling) and the process of mechanical work. The rate of gain or loss in the stored energy of a system is determined by the rates of these two processes. In open systems, the flow of matter is another energy transfer mechanism, and extra terms must be included in the expression of the first law.
The First Law clarifies the nature of energy. It is a stored quantity which is independent of any particular process path, i.e., it is independent of the system history. If a system undergoes a thermodynamic cycle, whether it becomes warmer, cooler, larger, or smaller, then it will have the same amount of energy each time it returns to a particular state. Mathematically speaking, energy is a state function and infinitesimal changes in the energy are exact differentials.
All laws of thermodynamics but the First are statistical and simply describe the tendencies of macroscopic systems. For microscopic systems with few particles, the variations in the parameters become larger than the parameters themselves, and the assumptions of thermodynamics become meaningless. The First Law, i.e. the law of conservation, has become the most secure of all basic laws of science. At present, it is unquestioned.[citation needed]
[edit] Fundamental Thermodynamic Relation
The first law can be expressed as the Fundamental Thermodynamic Relation:
Here, E is internal energy, T is temperature, S is entropy, p is pressure, and V is volume. This is a statement of conservation of energy: The net change in internal energy (dE) equals the heat energy that flows in (TdS), minus the energy that flows out via the system performing work (pdV).
[edit] Second law
The entropy of an isolated system not in equilibrium will tend to increase over time, approaching a maximum value at equilibrium.
In a simple manner, the second law states that "energy systems have a tendency to increase their entropy" rather than decrease it. This can also be stated as "heat can spontaneously flow from a higher-temperature region to a lower-temperature region, but not the other way around." (Heat can flow from cold to hot, but not spontaneously—for example, a refrigerator requires electricity.)
A way of looking at the second law for non-scientists is to look at entropy as a measure of disorder. So, for example, a broken cup has less order than an intact one. Likewise, solid crystals, the most organized form of matter, have very low entropy values; and gases, which are highly disorganized, have high entropy values.
The entropy of a thermally isolated macroscopic system never decreases. However, a microscopic system may exhibit fluctuations of entropy opposite to that dictated by the Second Law (see Maxwell's demon and Fluctuation Theorem).
[edit] Third law
As temperature approaches absolute zero, the entropy of a system approaches a constant minimum.
In brief, this postulates that entropy is temperature dependent and leads to the formulation of the idea of absolute zero.
[edit] Tentative fourth laws or principles
Over the years, various thermodynamic researchers have come forward to ascribe to or to postulate potential fourth laws of thermodynamics (either suggesting that a widely-accepted principle should be called the fourth law, or proposing entirely new laws); in some cases, even fifth or sixth laws of thermodynamics are proposed[1]. Most fourth law statements, however, are speculative and far from agreed upon.
The most commonly proposed Fourth Law is the Onsager reciprocal relations, which give a quantitative relation between the parameters of a system in which heat and matter are simultaneously flowing.
Other tentative fourth law statements are attempts to apply thermodynamics to evolution. In the late 19th century, thermodynamicist Ludwig Boltzmann argued that the fundamental object of contention in the life-struggle in the evolution of the organic world is 'available energy'. Another example is the maximum power principle as put forward initially by biologist Alfred Lotka in his 1922 article Contributions to the Energetics of Evolution.[2] Most variations of hypothetical fourth laws (or principles) have to do with the environmental sciences, biological evolution, or galactic phenomena.[3]
[edit] History
The first established principle which eventually became the Second Law was formulated by Sadi Carnot in 1824. By 1860, as found in the works of those such as Rudolf Clausius and William Thomson, there were two established "principles" of thermodynamics, the first principle and the second principle. As the years passed, these principles turned into "laws." By 1873, for example, thermodynamicist Josiah Willard Gibbs, in his “Graphical Methods in the Thermodynamics of Fluids”, clearly stated that there were two absolute laws of thermodynamics, a first law and a second law.
[edit] See also
- Conservation law
- Heat death of the universe
- Laws of science
- Philosophy of thermal and statistical physics
- Table of thermodynamic equations
[edit] References
- ^ A Proposed 5th Law of Thermodynamics
- ^ A.J.Lotka (1922a) 'Contribution to the energetics of evolution' [PDF]. Proc Natl Acad Sci, 8: pp. 147–51.
- ^ Morel, R.E. ,Fleck, George. (2006). "Fourth Law of Thermodynamics" Chemistry, Vol. 15, Iss. 4
[edit] Further reading
- Goldstein, Martin, and Inge F., 1993. The Refrigerator and the Universe. Harvard Univ. Press. A gentle introduction.