Coeliac disease
From Wikipedia, the free encyclopedia
| Coeliac disease Classification and external resources |
|
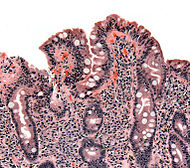
| Biopsy of small bowel showing coeliac disease manifested by blunting of villi, crypt hyperplasia, and lymphocyte infiltration of crypts. | |
| ICD-10 | K90.0 |
| ICD-9 | 579.0 |
| OMIM | 212750 |
| DiseasesDB | 2922 |
| MedlinePlus | 000233 |
| eMedicine | med/308 ped/2146 radio/652 |
| MeSH | D002446 |
Cœliac disease (pronounced /ˈsiːli.æk/), also spelled celiac disease, is an autoimmune disorder of the small intestine that occurs in genetically predisposed people of all ages from middle infancy on up. Symptoms include chronic diarrhœa, failure to thrive (in children), and fatigue, but these may be absent and symptoms in all other organ systems have been described. A growing portion of diagnoses are being made in asymptomatic persons as a result of increased screening.[1]
Coeliac disease is caused by a reaction to gliadin, a gluten protein found in wheat (and similar proteins of the tribe Triticeae which includes other cultivars such as barley and rye). Upon exposure to gliadin, the enzyme tissue transglutaminase modifies the protein, and the immune system cross-reacts with the small bowel tissue, causing an inflammatory reaction. That leads to flattening of the lining of the small intestine (called villous atrophy). This interferes with the absorption of nutrients because the intestinal villi are responsible for absorption. The only effective treatment is a lifelong gluten-free diet. While the disease is caused by a reaction to wheat proteins, it is not the same as wheat allergy.
This condition has several other names, including: cœliac disease (with "œ" ligature), c(o)eliac sprue, non-tropical sprue, endemic sprue, gluten enteropathy or gluten-sensitive enteropathy, and gluten intolerance. The term coeliac derives from the Greek κοιλιακός (koiliakόs, abdominal), and was introduced in the 19th century in a translation of what is generally regarded as an ancient Greek description of the disease by Aretaeus of Cappadocia.[2]
Contents |
[edit] Signs and symptoms
Classic symptoms of coeliac disease include diarrhoea, weight loss (or stunted growth in children), and fatigue, but while coeliac disease is primarily a bowel disease, bowel symptoms may also be limited or even absent. Some patients are diagnosed with symptoms related to the decreased absorption of nutrients or with various symptoms which, although statistically linked, have no clear relationship with the malfunctioning bowel. Given this wide range of possible symptoms, the classic triad is no longer a requirement for diagnosis.
Children between 9 and 24 months tend to present with bowel symptoms and growth problems shortly after first exposure to gluten-containing products. Older children may have more malabsorption-related problems and psychosocial problems, while adults generally have malabsorptive problems.[3] Many adults with subtle disease only have fatigue or anaemia.[1]
[edit] Gastrointestinal
The diarrhoea characteristic of coeliac disease is pale, voluminous and malodorous. Abdominal pain and cramping, bloatedness with abdominal distention (thought to be due to fermentative production of bowel gas) and mouth ulcers[4] may be present. As the bowel becomes more damaged, a degree of lactose intolerance may develop. However, the variety of gastrointestinal symptoms that may be present in patients with coeliac disease is great, and some may have a normal bowel habit or even tend towards constipation. Frequently the symptoms are ascribed to irritable bowel syndrome (IBS), only later to be recognised as coeliac disease; a small proportion of patients with symptoms of IBS have underlying coeliac disease, and screening may be justified.[5]
Coeliac disease leads to an increased risk of both adenocarcinoma and lymphoma of the small bowel, which returns to baseline with diet. Longstanding disease may lead to other complications, such as ulcerative jejunitis (ulcer formation of the small bowel) and stricturing (narrowing as a result of scarring).[6]
[edit] Malabsorption-related
The changes in the bowel make it less able to absorb nutrients, minerals and the fat-soluble vitamins A, D, E, and K.[3]
- The inability to absorb carbohydrates and fats may cause weight loss (or failure to thrive/stunted growth in children) and fatigue or lack of energy.
- Anaemia may develop in several ways: iron malabsorption may cause iron deficiency anaemia, and folic acid and vitamin B12 malabsorption may give rise to megaloblastic anaemia.
- Calcium and vitamin D malabsorption (and compensatory secondary hyperparathyroidism) may cause osteopenia (decreased mineral content of the bone) or osteoporosis (bone weakening and risk of fragility fractures).
- A small proportion (10%) have abnormal coagulation due to deficiency of vitamin K, and are slightly at risk for abnormal bleeding.
- Coeliac disease is also associated with bacterial overgrowth of the small intestine, which can worsen malabsorption, or cause malabsorption after treatment.[7]
[edit] Miscellaneous
Coeliac disease has been linked with a number of conditions. In many cases it is unclear whether the gluten-induced bowel disease is a causative factor or whether these conditions share a common predisposition.
- IgA deficiency is present in 2% of patients with coeliac disease, and in turn this condition features a tenfold increased risk of coeliac disease.[8][9] Other features of this condition are an increased risk of infections and autoimmune disease.
- Dermatitis herpetiformis; this itchy cutaneous condition has been linked to a transglutaminase enzyme in the skin, features small bowel changes identical to those in coeliac disease[10] and occurs more often (in 2%) in patients with coeliac disease.[3]
- Neurological associations: epilepsy, ataxia (coordination problems), myelopathy, peripheral neuropathy, and schizophrenia [11] have all been linked with coeliac disease, but the strength of these associations and the causality are still subject to debate.[12]
- Growth failure and/or pubertal delay in later childhood can occur even without obvious bowel symptoms or severe malnutrition. Evaluation of growth failure often includes coeliac screening.
- Miscarriage and infertility.
- Hyposplenism (a small and underactive spleen) - it is unclear whether this actually increases infection risk in the same way as in other people without a functioning spleen.[13]
- Other auto-immune disorders: diabetes mellitus type 1,[14] autoimmune thyroiditis,[15] primary biliary cirrhosis,[16] and microscopic colitis.[17]
[edit] Other grains
Wheat varieties or subspecies containing gluten and related species such as barley and rye also induce symptoms of coeliac disease.[18] A small minority of coeliac patients also react to oats.[19][20] It is most probable that oats produce symptoms due to cross contamination with other grains in the fields or in the distribution channels.[3] Generally, oats are therefore not recommended[18], though gluten-free oats are available in some locales and may be tried with caution.[21] Other cereals, such as maize (corn), quinoa, millet, sorghum, chia seed, and rice are safe for patients to consume. Non-cereal carbohydrate-rich foods such as potatoes and bananas do not contain gluten and do not trigger symptoms.
[edit] Diagnosis
There are several tests that can be used to assist in diagnosis. The level of symptoms may determine the order of the tests, but all tests lose their usefulness if the patient is already taking a gluten-free diet. Intestinal damage begins to heal within weeks of gluten being removed from the diet, and antibody levels decline over months. For those who have already started on a gluten-free diet, it may be necessary to perform a re-challenge with 10 g of gluten (four slices of bread) per day over 2–6 weeks before repeating the investigations. Those who experience severe symptoms (e.g. diarrhoea) earlier can be regarded as sufficiently challenged and can be tested earlier.[3]
Combining findings into a prediction rule to guide use of endoscopy reported a sensitivity of 100% (it would identify all the cases) and specificity of 61% (it would be incorrectly positive in 39%). The prediction rule recommends that patients with high risk symptoms or positive serology should undergo endoscopy. The study defined high risk symptoms as weight loss, anaemia (haemoglobin less than 120 g/l in females and less than 130 g/l in males), or diarrhoea (more than three loose stools per day).[22]
[edit] Blood tests
[edit] Antibody testing
Serological blood tests are the first-line investigation required to make a diagnosis of coeliac disease. Serology for anti-tTG antibodies has superseded older serological tests and has a high sensitivity (99%) and specificity (>90%) for identifying coeliac disease. Modern anti-tTG assays rely on a human recombinant protein as an antigen.[23]
Because of the major implications of a diagnosis of coeliac disease, professional guidelines recommend that a positive blood test is still followed by an endoscopy/gastroscopy and biopsy. A negative serology test may still be followed by a recommendation for endoscopy and duodenal biopsy if clinical suspicion remains high due to the 1 in 100 "false-negative" result. As such tissue biopsy is still considered the gold standard in the diagnosis of coeliac disease.[3][6]
Historically three other antibodies were measured: anti-reticulin (ARA), anti-gliadin (AGA) and anti-endomysium (EMA) antibodies. Serology may be unreliable in young children, with anti-gliadin performing somewhat better than other tests in children under five.[24] Serology tests are based on indirect immunofluorescence (reticulin, gliadin and endomysium) or ELISA (gliadin or tissue transglutaminase, tTG).[25]
Guidelines recommend that a total serum IgA level is checked in parallel, as coeliac patients with IgA deficiency may be unable to produce the antibodies on which these tests depend ("false negative"). In those patients, IgG antibodies against transglutaminase (IgG-tTG) may be diagnostic.[26]
| Test | sensitivity | specificity |
|---|---|---|
| HLA-DQ2 | 94% | 73% |
| HLA-DQ8 | 12% | 81% |
[edit] HLA genetic typing
Antibody testing and HLA testing have similar accuracies.[27]
[edit] Endoscopy

An upper endoscopy with biopsy of the duodenum (beyond the duodenal bulb) or jejunum is performed. It is important for the physician to obtain multiple samples (four to eight) from the duodenum. Not all areas may be equally affected; if biopsies are taken from healthy bowel tissue, the result would be a false negative.[6]
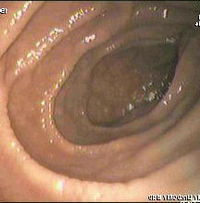
Most patients with coeliac disease have a small bowel that appears normal on endoscopy; however, five concurrent endoscopic findings have been associated with a high specificity for coeliac disease: scalloping of the small bowel folds (pictured), paucity in the folds, a mosaic pattern to the mucosa (described as a cracked-mud appearance), prominence of the submucosa blood vessels, and a nodular pattern to the mucosa.[28]
Until the 1970s, biopsies were obtained using metal capsules attached to a suction device. The capsule was swallowed and allowed to pass into the small intestine. After x-ray verification of its position, suction was applied to collect part of the intestinal wall inside the capsule. One often-utilised capsule system is the Watson capsule. This method has now been largely replaced by fibre-optic endoscopy, which carries a higher sensitivity and a lower frequency of errors.[29]
[edit] Pathology
The classic pathology changes of coeliac disease in the small bowel are categorised by the "Marsh classification":[30]
- Marsh stage 0: normal mucosa
- Marsh stage 1: increased number of intra-epithelial lymphocytes, usually exceeding 20 per 100 enterocytes
- Marsh stage 2: proliferation of the crypts of Lieberkuhn
- Marsh stage 3: partial or complete villous atrophy
- Marsh stage 4: hypoplasia of the small bowel architecture
The changes classically improve or reverse after gluten is removed from the diet, so many official guidelines recommend a repeat biopsy several (4–6) months after commencement of gluten exclusion.[3]
In some cases a deliberate gluten challenge, followed by biopsy, may be conducted to confirm or refute the diagnosis. A normal biopsy and normal serology after challenge indicates the diagnosis may have been incorrect.[3] Patients are warned that one does not "outgrow" coeliac disease in the same way as childhood food intolerances.
[edit] Other diagnostic tests
Other tests that may assist in the diagnosis are blood tests for a full blood count, electrolytes, calcium, renal function, liver enzymes, vitamin B12 and folic acid levels. Coagulation testing (prothrombin time and partial thromboplastin time) may be useful to identify deficiency of vitamin K, which predisposes patients to hemorrhage. These tests should be repeated on follow-up, as well as anti-tTG titres.[3]
Some professional guidelines[3] recommend screening of all patients for osteoporosis by DXA/DEXA scanning.
[edit] Pathophysiology
Coeliac disease appears to be polyfactorial, both in that more than one genetic factor can cause the disease and also more than one factor is necessary for the disease to manifest in a patient.
Almost all coeliac patients have the variant HLA DQ2 allele.[1] However, about 20–30% of people without coeliac disease have inherited an HLA-DQ2 allele.[27] This suggests additional factors are needed for coeliac disease to develop. Furthermore, about 5% of those people who do develop coeliac disease do not have the DQ2 gene.[1]
The HLA-DQ2 allele shows incomplete penetrance, as the gene alleles associated with the disease appear in most patients, but are neither present in all cases nor sufficient by themselves to cause the disease.
[edit] Genetics
The vast majority of coeliac patients have one of two types of HLA DQ.[27] This gene is part of the MHC class II antigen-presenting receptor (also called the human leukocyte antigen) system and distinguishes cells between self and non-self for the purposes of the immune system. The gene is located on the short arm of the sixth chromosome, and as a result of the linkage this locus has been labeled CELIAC1.
There are 7 HLA DQ variants (DQ2 and DQ4 through DQ9). Over 95% of coeliac patients have the isoform of DQ2 or DQ8, which is inherited in families. The reason these genes produce an increase in risk of coeliac disease is that the receptors formed by these genes bind to gliadin peptides more tightly than other forms of the antigen-presenting receptor. Therefore, these forms of the receptor are more likely to activate T lymphocytes and initiate the autoimmune process.[1]
Most coeliac patients bear a two-gene HLA-DQ2 haplotype referred to as DQ2.5 haplotype. This haplotype is composed of 2 adjacent gene alleles, DQA1*0501 and DQB1*0201, which encode the two subunits, DQ α5 and DQ β2. In most individuals, this DQ2.5 isoform is encoded by one of two chromosomes 6 inherited from parents. Most coeliacs inherit only one copy of this DQ2.5 haplotype, while some inherit it from both parents; the latter are especially at risk for coeliac disease, as well as being more susceptible to severe complications.[32] Some individuals inherit DQ2.5 from one parent and portions of the haplotype (DQB1*02 or DQA1*05) from the other parent, increasing risk. Less commonly, some individuals inherit the DQA1*05 allele from one parent and the DQB1*02 from the other parent, called a trans-haplotype association, and these individuals are at similar risk for coeliac disease as those with a single DQ2.5 bearing chromosome 6, but in this instance disease tends not to be familial. Among the 6% of European coeliacs that do not have DQ2.5(cis or trans) or DQ8 (encoded by the haplotype DQA1*03:DQB1*0302), 4% have the DQ2.2 isoform and the remaining 2% lack DQ2 or DQ8.[33]
The frequency of these genes varies geographically. DQ2.5 has high frequency in peoples of North and Western Europe (Basque Country, Ireland,[34] with highest frequencies), portions of Africa, and is associated with disease in India,[35] but is not found along portions of the West Pacific rim. DQ8, spread more globally than DQ2.5, is more prevalent from South and Central America (up to 90% phenotype frequency).[36]
[edit] Prolamins
The majority of the proteins in food responsible for the immune reaction in coeliac disease are the prolamins. These are storage proteins rich in proline (prol-) and glutamine (-amin) that dissolve in alcohols and are resistant to proteases and peptidases of the gut.[37][1] One region of α-gliadin stimulates membrane cells, enterocytes, of the intestine to allow larger molecules around the sealant between cells. Disruption of tight junctions allow peptides larger than 3 amino acids to enter circulation.[38]

Membrane leaking permits peptides of gliadin that stimulate two levels of immune response, the innate response and the adaptive (T-helper cell mediated) response. One protease resistant peptide from α-gliadin contains a region that stimulates lymphocytes and results in the release of interleukin-15. This innate response to gliadin results in immune system signalling that attracts inflammatory cells and increases the release inflammatory chemicals.[1] The strongest and most common adaptive response to gliadin is directed toward a α2-gliadin fragment of 33 amino acids in length.[1] The response to 33mer occurs in most coeliacs who have a DQ2 isoform. This peptide, when altered by intestinal transglutaminase, has a high density of overlapping T-cell epitopes. This increases the likelihood that the DQ2 isoform will bind and stay bound to peptide when recognised by T-cells.[39] Gliadin in wheat is the best-understood member of this family, but other prolamins exist and hordein (from barley), and secalin (from rye) may contribute to coeliac disease.[1][40] However, not all prolamins will cause this immune reaction and there is ongoing controversy on the ability of avenin (the prolamin found in oats) to induce this response in coeliac disease.
[edit] Tissue transglutaminase
Anti-transglutaminase antibodies to the enzyme tissue transglutaminase (tTG) are found in an overwhelming majority of cases.[41] Tissue transglutaminase modifies gluten peptides into a form that may stimulate the immune system more effectively.[1]
Stored biopsies from suspected coeliac patients has revealed that autoantibody deposits in the subclinical coeliacs are detected prior to clinical disease. These deposits are also found in patients who present with other autoimmune diseases, anaemia or malabsorption phenomena at a much increased rate over the normal population.[42] Endomysial component of antibodies (EMA) to tTG are believed to be directed toward cell surface transglutaminase, and these antibodies are still used in confirming a coeliac disease diagnosis. However, a 2006 study showed that EMA-negative coeliac patients tend to be older males with more severe abdominal symptoms and a lower frequency of "atypical" symptoms including autoimmune disease.[43] In this study the anti-tTG antibody deposits did not correlate with the severity of villous destruction. These findings, coupled with recent work showing that gliadin has an innate response component,[44] suggests that gliadin may be more responsible for the primary manifestations of coeliac disease whereas tTG is a bigger factor in secondary effects such as allergic responses and secondary autoimmune diseases. In a large percentage of coeliac patients the anti-tTG antibodies also recognise a rotavirus protein called VP7. These antibodies stimulate monocytes proliferation and rotavirus infection might explain some early steps in the cascade of immune cell proliferation.[45] Indeed, earlier studies of rotavirus damage in the gut showed this causes a villous atrophy.[46] This suggests that viral proteins may take part in the initial flattening and stimulate self-crossreactive anti-VP7 production. Antibodies to VP7 may also slow healing until the gliadin mediated tTG presentation provides a second source of crossreactive antibodies.
[edit] Villous atrophy and malabsorption
The inflammatory process, mediated by T cells, leads to disruption of the structure and function of the small bowel's mucosal lining, and causes malabsorption as it impairs the body's ability to absorb nutrients, minerals and fat-soluble vitamins A, D, E and K from food. Lactose intolerance may be present due to the decreased bowel surface and reduced production of lactase but typically resolves once the condition is treated.
Alternative causes of this tissue damage have been proposed and involve release of interleukin 15 and activation of the innate immune system by a shorter gluten peptide (p31–43/49). This would trigger killing of enterocytes by lymphocytes in the epithelium.[1] The villous atrophy seen on biopsy may also be due to unrelated causes, such as tropical sprue, giardiasis and radiation enteritis. While positive serology and typical biopsy are highly suggestive of coeliac disease, lack of response to diet may require these alternative diagnoses to be considered.[6]
[edit] Risk modifiers
There are various theories as to what determines whether a genetically susceptible individual will go on to develop coeliac disease. Major theories include infection by rotavirus[47] or human intestinal adenovirus.[48] Some research has suggested that smoking is protective against adult onset coeliac disease.[49]
A 2005 prospective and observational study found that timing of the exposure to gluten in childhood was an important risk modifier. People exposed to wheat, barley, or rye before the gut barrier has fully developed (within the first three months after birth) had five times the risk of developing coeliac disease relative to those exposed at between 4 to 6 months after birth. Those exposed even later than six months after birth, were found to have only a slightly increased risk relative to those exposed at between 4 to 6 months after birth.[50] However a 2006 study with similar numbers found just the reverse, that early introduction of grains was protective.[51] Breastfeeding may also reduce risk. A meta-analysis indicates that prolonging breastfeeding until the introduction of gluten-containing grains into the diet was associated with a 52% reduced risk of developing coeliac disease in infancy; whether this persists into adulthood is not clear.[52]
[edit] Treatment
[edit] Diet
At present, the only effective treatment is a life-long gluten-free diet.[18] No medication exists that will prevent damage, or prevent the body from attacking the gut when gluten is present. Strict adherence to the diet allows the intestines to heal, leading to resolution of all symptoms in most cases and, depending on how soon the diet is begun, can also eliminate the heightened risk of osteoporosis and intestinal cancer.[53] Dietician input is generally requested to ensure the patient is aware which foods contain gluten, which foods are safe, and how to have a balanced diet despite the limitations. In many countries gluten-free products are available on prescription and may be reimbursed by health insurance plans. More manufacturers are producing gluten-free products, some of which are almost indistinguishable from their gluten-containing counterparts.
The diet can be cumbersome; failure to comply with the diet may cause relapse. The term "gluten-free" is generally used to indicate a supposed harmless level of gluten rather than a complete absence.[54] The exact level at which gluten is harmless is uncertain and controversial. A recent systematic review tentatively concluded that consumption of less than 10 mg of gluten per day is unlikely to cause histological abnormalities, although it noted that few reliable studies had been done.[54] Regulation of the label "gluten-free" varies widely by country. For example, in the United States the term "gluten-free" is not yet regulated.[55] The current international Codex Alimentarius standard, established in 1981, allows for 50 mg N/100 g on dry matter,[56] although a proposal for a revised standard of 20 ppm in naturally gluten-free products and 200 ppm in products rendered gluten-free has been accepted.[57] Gluten-free products are usually more expensive and harder to find than common gluten-containing foods.[58] Since ready-made products often contain traces of gluten, some coeliacs may find it necessary to cook from scratch.[57]
Even while on a diet, health-related quality of life (HRQOL) may be lower in people with coeliac disease. Studies in the United States have found that quality of life becomes comparable to the general population after staying on the diet while studies in Europe have found that quality of life remains lower, although the surveys are not quite the same.[59] Men tend to report more improvement than women.[60] Some have persisting digestive symptoms or dermatitis herpetiformis, mouth ulcers, osteoporosis and resultant fractures. Symptoms suggestive of irritable bowel syndrome may be present, and there is an increased rate of anxiety, fatigue, dyspepsia and musculoskeletal pain.[61]
[edit] Refractory disease
A tiny minority of patients suffer from refractory disease, which means they do not improve on a gluten-free diet. This may be because the disease has been present for so long that the intestines are no longer able to heal on diet alone, or because the patient is not adhering to the diet, or because the patient is consuming foods that are inadvertently contaminated with gluten. If alternative causes have been eliminated, steroids or immunosuppressants (such as azathioprine) may be considered in this scenario.[6]
[edit] Experimental treatments
Various other approaches are being studied that would reduce the need of dieting. All are still under development, and are not expected to be available to the general public for a while:[1]
- Genetically engineered wheat species, or wheat species that have been selectively bred to be minimally immunogenic. This, however, could interfere with the effects that gliadin has on the quality of dough.
- A combination of enzymes (prolyl endopeptidase and a barley glutamine-specific cysteine endopeptidase (EP-B2)) that degrade the putative 33-mer peptide in the duodenum. This combination would enable coeliac disease patients to consume gluten-containing products.[62]
- Inhibition of zonulin, an endogenous signalling protein linked to increased permeability of the bowel wall and hence increased presentation of gliadin to the immune system.[63]
- Other treatments aimed at other well-understood steps in the pathogenesis of coeliac disease, such as the action of HLA-DQ2 or tissue transglutaminase and the MICA/NKG2D interaction that may be involved in the killing of enterocytes (bowel lining cells).
[edit] Screening and case finding
There is significant debate as to the benefits of screening. Some studies suggest that early detection would decrease the risk of osteoporosis and anaemia. In contrast, a cohort studied in Cambridge suggested that people with undetected coeliac disease had a beneficial risk profile for cardiovascular disease (less overweight, lower cholesterol levels).[1]
Due to its high sensitivity, serology has been proposed as a screening measure, because the presence of antibodies would detect previously undiagnosed cases of coeliac disease and prevent its complications in those patients. Serology may also be used to monitor adherence to diet: in those who still ingest gluten, antibody levels remain elevated.[3][6]
In the United Kingdom, the National Institute for Health and Clinical Excellence (NICE) recommends screening for coeliac disease in patients with newly diagnosed chronic fatigue syndrome[64] and irritable bowel syndrome.[65]
Other clinical scenarios in which screening may be justified include type 1 diabetes,[14] unexplained iron-deficiency anaemia,[66][67] Down's syndrome, Turner's syndrome, lupus, and autoimmune thyroid disease.[68]
[edit] Epidemiology
The prevalence of clinically diagnosed disease (symptoms prompting diagnostic testing) is 0.05–0.27% in various studies. However, population studies from parts of Europe, India, South America, Australasia and the USA (using serology and biopsy) indicate that the prevalence may be between 0.33 and 1.06% in children (5.66% in one study of Sahrawi children[69]) and 0.18–1.2% in adults.[1] People of African, Japanese and Chinese descent are rarely diagnosed; this reflects a much lower prevalence of the genetic risk factors. Population studies also indicate that a large proportion of coeliacs remain undiagnosed; this is due to many clinicians being unfamiliar with the condition.[70]
A large multicentre study in the U.S. found a prevalence of 0.75% in not-at-risk groups, rising to 1.8% in symptomatic patients, 2.6% in second-degree relatives of a patient with coeliac disease and 4.5% in first-degree relatives. This profile is similar to the prevalence in Europe.[71] Other populations at increased risk for coeliac disease, with prevalence rates ranging from 5% to 10%, include individuals with Down and Turner syndromes, type 1 diabetes, and autoimmune thyroid disease, including both hyperthyroidism (overactive thyroid) and hypothyroidism (underactive thyroid).[72]
Historically, coeliac disease was thought to be rare, with a prevalence of about 0.02%.[72] Recent increases in the number of reported cases may be due to changes in diagnostic practice.[73]
[edit] Social and religious issues
[edit] Christian churches & the Eucharist
Most mainstream Christian churches offer their communicants gluten-free alternatives to the sacramental bread, usually in the form of a rice-based cracker or gluten-free bread. These include United Methodist, Christian Reformed, Episcopal, Lutheran, The Church of Jesus Christ of Latter-day Saints, and many others.[74]
[edit] Roman Catholic position
Roman Catholic doctrine states that for a valid Eucharist the bread must be made from wheat. In 2002, the Congregation for the Doctrine of the Faith approved German-made low-gluten hosts, which meet all of the Catholic Church's requirements, for use in Italy; although not entirely gluten-free, they were also approved by the Italian Celiac Association.[75] Some Catholic coeliac sufferers have requested permission to use rice wafers; such petitions have always been denied.[76] The issue is more complex for priests. Though a Catholic (lay or ordained) receiving under either form is receiving Christ "whole and entire," his body, blood, soul, and divinity, the priest, who is acting in persona Christi, is required to receive under both species when offering Mass — not for the validity of his Communion, but for the fullness of the sacrifice of the Mass. On 22 August 1994, the Congregation for the Doctrine of the Faith apparently barred coeliacs from ordination, stating, "Given the centrality of the celebration of the Eucharist in the life of the priest, candidates for the priesthood who are affected by coeliac disease or suffer from alcoholism or similar conditions may not be admitted to holy orders." After considerable debate, the congregation softened the ruling on 24 July 2003 to "Given the centrality of the celebration of the Eucharist in the life of a priest, one must proceed with great caution before admitting to Holy Orders those candidates unable to ingest gluten or alcohol without serious harm."[77]
As of January 2004, an extremely low-gluten host became available in the United States. The Benedictine Sisters of Perpetual Adoration in Clyde, MO produce low-gluten host safe for celiacs and also approved by the Catholic Church for use at Mass. The hosts are made and packaged in a dedicated wheat-free / gluten-free environment. Gluten content analysis found no detectable amount of gluten, though the reported gluten content is 0.01% as that was the lowest limit of detection possible with the utilized analysis technique. In an article from the Catholic Review (15 February 2004) Dr. Alessio Fasano was quoted as declaring these hosts "perfectly safe for celiac sufferers." [78]
[edit] Coeliacs and Passover
The Jewish festival of Pesach (Passover) may present problems with its obligation to eat matzo, which is unleavened bread made in a strictly controlled manner from wheat, barley, spelt, oats, or rye. This rules out many other grains that are normally used as substitutes for people with gluten sensitivity, especially for Ashkenazi Jews who also avoid rice. Many kosher for Passover products avoid grains altogether and are therefore gluten-free. Potato starch is the primary starch used to replace the grains. Consuming matzo is mandatory on the first night of Pesach only. Jewish law holds that a person should not seriously endanger one's health in order to fulfill a commandment. Thus, a person with severe coeliac disease is not required, or even allowed, to eat any matzo other than gluten-free matzo. The most commonly used gluten-free matzo is made from oats.[79]
[edit] History
Aretaeus of Cappadocia, living in the second century, recorded a malabsorptive syndrome with chronic diarrhoea. His "Cœliac Affection" (coeliac from Greek κοιλιακός koiliakos, abdominal) gained the attention of Western medicine when Francis Adams presented a translation of Aretaeus' work at the Sydenham Society in 1856. The patient had stomach pain and was atrophied, pale, feeble and incapable of work. The diarrhoea manifested as loose stools that were white, malodorous and flatulent and the disease was intractable and liable to periodic return. The problem, Aretaeus believed, was a lack of heat in the stomach necessary to digest the food and a reduced ability to distribute the digestive products throughout the body, this incomplete digestion resulting in the diarrhoea, He regarded this as an affliction of the old and more commonly affecting women, explicitly excluding children. The cause, according to Aretaeus, was sometimes either another chronic disease or even consuming "a copious draught of cold water".[2]
The paediatrician Samuel Gee gave the first modern-day description of the condition in a lecture at Hospital for Sick Children, Great Ormond Street, London in 1887. Gee acknowledged earlier descriptions and terms for the disease and adopted the same term as Aretaeus (coeliac disease). Unlike Aretaeus, he included children in the scope of the affliction, particularly those between one and five years old. Gee found the cause to be obscure and failed to spot anything abnormal during post-mortem examination (the lining of the small bowel quickly deteriorates on death).[80] He perceptively stated "if the patient can be cured at all, it must be by means of diet." Gee recognised that milk intolerance is a problem with coeliac children and that highly starched foods should be avoided. However, he forbade rice, sago, fruit and vegetables, which all would have been safe to eat and he recommended raw meat as well as thin slices of toasted bread. Gee highlighted particular success with a child "who was fed upon a quart of the best Dutch mussels daily". However, the child could not bear this diet for more than one season.[81]
Christian Archibald Herter, an American physician, wrote a book in 1908 on children with coeliac disease, which he called "intestinal infantilism". He noted their growth was retarded and that fat was better tolerated than carbohydrate. The eponym Gee-Herter disease was sometimes used to acknowledge both contributions.[82][83] Sydney V. Haas, an American paediatrician, reported positive effects of a diet of bananas in 1924.[84] This diet remained in vogue until the actual cause of coeliac disease was determined.
While a role for carbohydrates had been suspected, the link with wheat was not made until the 1940s by the Dutch paediatrician Dr Willem Dicke.[85] It is likely that clinical improvement of his patients during the Dutch famine of 1944 (during which flour was sparse) may have contributed to his discovery.[86] The link with the gluten component of wheat was made in 1952 by a team from Birmingham, England.[87] Villous atrophy was described by British physician John W. Paulley in 1954.[88] Paulley was able to examine biopsies taken from patients during abdominal operations.[80] Dr Margo Shiner, working on Prof Sheila Sherlock's team at the Postgraduate Medical School in London, described the principles of small bowel biopsy in 1956.[89]
Throughout the 1960s other features of coeliac disease were elucidated. Its hereditary character was recognised in 1965.[90] In 1966 dermatitis herpetiformis was linked to gluten sensitivity.[10]
[edit] References
- ^ a b c d e f g h i j k l m n van Heel D, West J (2006). "Recent advances in coeliac disease". Gut 55 (7): 1037–46. doi:. PMID 16766754. http://gut.bmjjournals.com/cgi/content/full/55/7/1037.
- ^ a b Adams F, translator (1856). "On The Cœliac Affection". The extant works of Aretaeus, The Cappadocian. London: Sydenham Society. http://web.archive.org/web/20070311164628/http://www.chlt.org/sandbox/dh/aretaeusEnglish/page.102.a.php. Retrieved on 2006-09-04. See also Google Books entry
- ^ a b c d e f g h i j k Ciclitira, P (2002). "Interim Guidelines for the Management of Patients with Coeliac Disease". British Society of Gastroenterology. http://www.bsg.org.uk/bsgdisp1.php?id=c9c5177d2b91e3228066. Retrieved on 2007-03-07.
- ^ Ferguson R, Basu M, Asquith P, Cooke W (1976). "Jejunal mucosal abnormalities in patients with recurrent aphthous ulceration". Br Med J 1 (6000): 11–13. PMID 1247715.
- ^ Spiegel BM, DeRosa VP, Gralnek IM, Wang V, Dulai GS (June 2004). "Testing for celiac sprue in irritable bowel syndrome with predominant diarrhea: a cost-effectiveness analysis". Gastroenterology 126 (7): 1721–32. doi:. PMID 15188167.
- ^ a b c d e f "American Gastroenterological Association medical position statement: Celiac Sprue". Gastroenterology 120 (6): 1522–5. 2001. PMID 11313323. http://www.gastrojournal.org/article/PIIS0016508501701618/fulltext.
- ^ Tursi A, Brandimarte G, Giorgetti G (2003). "High prevalence of small intestinal bacterial overgrowth in celiac patients with persistence of gastrointestinal symptoms after gluten withdrawal". Am J Gastroenterol 98 (4): 839–43. doi:. PMID 12738465.
- ^ Crabbé P, Heremans J (1967). "Selective IgA deficiency with steatorrhea. A new syndrome". Am J Med 42 (2): 319–26. doi:. PMID 4959869.
- ^ Collin P, Mäki M, Keyriläinen O, Hällström O, Reunala T, Pasternack A (1992). "Selective IgA deficiency and coeliac disease". Scand J Gastroenterol 27 (5): 367–71. doi:. PMID 1529270.
- ^ a b Marks J, Shuster S, Watson A (1966). "Small-bowel changes in dermatitis herpetiformis". Lancet 2 (7476): 1280–2. doi:. PMID 4163419.
- ^ Eaton W, Mortensen P, Esben A, Byrne M, Mors O, Ewald H (2002). "Coeliac disease and schizophrenia: population based case control study with linkage of Danish national registers". British Medical Journal 328: 438–9. doi:. PMID 14976100. PMC: 344262. http://www.bmj.com/cgi/content/short/328/7437/438.
- ^ Pengiran Tengah D, Wills A, Holmes G (2002). "Neurological complications of coeliac disease". Postgrad Med J 78 (921): 393–8. doi:. PMID 12151653. http://pmj.bmjjournals.com/cgi/content/full/78/921/393.
- ^ Ferguson A, Hutton M, Maxwell J, Murray D (1970). "Adult coeliac disease in hyposplenic patients". Lancet 1 (7639): 163–4. doi:. PMID 4189238.
- ^ a b Holmes G (2001). "Coeliac disease and Type 1 diabetes mellitus - the case for screening". Diabet Med 18 (3): 169–77. doi:. PMID 11318836.
- ^ Collin P, Kaukinen K, Välimäki M, Salmi J (2002). "Endocrinological disorders and celiac disease". Endocr Rev 23 (4): 464–83. doi:. PMID 12202461. http://edrv.endojournals.org/cgi/content/full/23/4/464.
- ^ Kingham J, Parker D (1998). "The association between primary biliary cirrhosis and coeliac disease: a study of relative prevalences". Gut 42 (1): 120–2. PMID 9518232. PMC: 1726939. http://gut.bmj.com/cgi/content/full/42/1/120.
- ^ Matteoni C, Goldblum J, Wang N, Brzezinski A, Achkar E, Soffer E (2001). "Celiac disease is highly prevalent in lymphocytic colitis". J Clin Gastroenterol 32 (3): 225–7. doi:. PMID 11246349.
- ^ a b c Kupper C (2005). "Dietary guidelines and implementation for celiac disease". Gastroenterology 128 (4 Suppl 1): S121–7. doi:. PMID 15825119.
- ^ Lundin K, Nilsen E, Scott H, Løberg E, Gjøen A, Bratlie J, Skar V, Mendez E, Løvik A, Kett K (2003). "Oats induced villous atrophy in coeliac disease". Gut 52 (11): 1649–52. doi:. PMID 14570737. PMC: 1773854. http://gut.bmjjournals.com/cgi/content/full/52/11/1649.
- ^ Størsrud S, Olsson M, Arvidsson Lenner R, Nilsson L, Nilsson O, Kilander A (2003). "Adult coeliac patients do tolerate large amounts of oats". Eur J Clin Nutr 57 (1): 163–9. doi:. PMID 12548312. http://www.nature.com/ejcn/journal/v57/n1/abs/1601525a.html.
- ^ "Canadian Celiac Association: Position Statement on Oats". http://www.celiac.ca/Articles/PABoats.html. Retrieved on 26 November 2008.
- ^ Hopper A, Cross S, Hurlstone D, McAlindon M, Lobo A, Hadjivassiliou M, Sloan M, Dixon S, Sanders D (2007). "Pre-endoscopy serological testing for coeliac disease: evaluation of a clinical decision tool". BMJ 334: 729. doi:. PMID 17383983. PMC: 1847864. http://www.bmj.com/cgi/content/full/334/7596/729.
- ^ Sblattero D, Berti I, Trevisiol C, et al (May 2000). "Human recombinant tissue transglutaminase ELISA: an innovative diagnostic assay for celiac disease". Am. J. Gastroenterol. 95 (5): 1253–7. doi:. PMID 10811336. http://www3.interscience.wiley.com/cgi-bin/fulltext/119001630/HTMLSTART.
- ^ Hill ID. "What are the sensitivity and specificity of serological tests for celiac disease? Do sensitivity and specificity vary in different populations?" In: NIH Consensus Development Conference on Celiac Disease. Bethesda, Md.: U.S. National Institutes of Health, 2004;27–31. PDF.
- ^ Wong R, Steele R, Reeves G, Wilson R, Pink A, Adelstein S (2003). "Antibody and genetic testing in coeliac disease". Pathology 35 (4): 285–304. PMID 12959764.
- ^ Korponay-Szabó I, Dahlbom I, Laurila K, Koskinen S, Woolley N, Partanen J, Kovács J, Mäki M, Hansson T (2003). "Elevation of IgG antibodies against tissue transglutaminase as a diagnostic tool for coeliac disease in selective IgA deficiency". Gut 52 (11): 1567–71. doi:. PMID 14570724. PMC: 1773847. http://gut.bmjjournals.com/cgi/content/full/52/11/1567.
- ^ a b c d Hadithi M, von Blomberg BM, Crusius JB, et al (2007). "Accuracy of serologic tests and HLA-DQ typing for diagnosing celiac disease". Ann. Intern. Med. 147 (5): 294–302. PMID 17785484. http://www.annals.org/cgi/content/full/147/5/294.
- ^ Niveloni S, Fiorini A, Dezi R, Pedreira S, Smecuol E, Vazquez H, Cabanne A, Boerr LA, Valero J, Kogan Z, Maurino E, Bai JC. (1998). "Usefulness of videoduodenoscopy and vital dye staining as indicators of mucosal atrophy of celiac disease: assessment of interobserver agreement". Gastrointestinal Endoscopy 47 (3): 223–229. doi:. PMID 9580349.
- ^ Mee A, Burke M, Vallon A, Newman J, Cotton P (1985). "Small bowel biopsy for malabsorption: comparison of the diagnostic adequacy of endoscopic forceps and capsule biopsy specimens". Br Med J (Clin Res Ed) 291 (6498): 769–72. PMID 3929934.
- ^ Marsh M (1992). "Gluten, major histocompatibility complex, and the small intestine. A molecular and immunobiologic approach to the spectrum of gluten sensitivity ('celiac sprue')". Gastroenterology 102 (1): 330–54. PMID 1727768.
- ^ Kim C, Quarsten H, Bergseng E, Khosla C, Sollid L (2004). "Structural basis for HLA-DQ2-mediated presentation of gluten epitopes in celiac disease". Proc Natl Acad Sci USA 101 (12): 4175–9. doi:. PMID 15020763. PMC: 384714. http://www.pnas.org/cgi/content/full/101/12/4175.
- ^ Jores RD, Frau F, Cucca F, et al (2007). "HLA-DQB1*0201 homozygosis predisposes to severe intestinal damage in celiac disease". Scand. J. Gastroenterol. 42 (1): 48–53. doi:. PMID 17190762.
- ^ Karell K, Louka AS, Moodie SJ, et al (2003). "HLA types in celiac disease patients not carrying the DQA1*05-DQB1*02 (DQ2) heterodimer: results from the European Genetics Cluster on Celiac Disease". Hum. Immunol. 64 (4): 469–77. doi:. PMID 12651074.
- ^ Michalski J, McCombs C, Arai T, Elston R, Cao T, McCarthy C, Stevens F (1996). "HLA-DR, DQ genotypes of celiac disease patients and healthy subjects from the West of Ireland". Tissue Antigens 47 (2): 127–33. PMID 8851726.
- ^ Kaur G, Sarkar N, Bhatnagar S, et al (2002). "Pediatric celiac disease in India is associated with multiple DR3-DQ2 haplotypes". Hum. Immunol. 63 (8): 677–82. doi:. PMID 12121676.
- ^ Layrisse Z, Guedez Y, Domínguez E, Paz N, Montagnani S, Matos M, Herrera F, Ogando V, Balbas O, Rodríguez-Larralde A (2001). "Extended HLA haplotypes in a Carib Amerindian population: the Yucpa of the Perija Range". Hum Immunol 62 (9): 992–1000. doi:. PMID 11543901.
- ^ Green PH, Cellier C (October 2007). "Celiac disease". N. Engl. J. Med. 357 (17): 1731–43. doi:. PMID 17960014.
- ^ Lammers KM, Lu R, Brownley J, et al (July 2008). "Gliadin induces an increase in intestinal permeability and zonulin release by binding to the chemokine receptor CXCR3". Gastroenterology 135 (1): 194–204.e3. doi:. PMID 18485912.
- ^ a b Qiao SW, Bergseng E, Molberg Ø, et al (August 2004). "Antigen presentation to celiac lesion-derived T cells of a 33-mer gliadin peptide naturally formed by gastrointestinal digestion". J. Immunol. 173 (3): 1757–62. PMID 15265905. http://www.jimmunol.org/cgi/pmidlookup?view=long&pmid=15265905.
- ^ Shan L, Qiao SW, Arentz-Hansen H, et al (2005). "Identification and analysis of multivalent proteolytically resistant peptides from gluten: implications for celiac sprue". J. Proteome Res. 4 (5): 1732–41. doi:. PMID 16212427.
- ^ Dieterich W, Ehnis T, Bauer M, Donner P, Volta U, Riecken E, Schuppan D (1997). "Identification of tissue transglutaminase as the autoantigen of celiac disease". Nat Med 3 (7): 797–801. doi:. PMID 9212111.
- ^ Kaukinen K, Peraaho M, Collin P, Partanen J, Woolley N, Kaartinen T, Nuuntinen T, Halttunen T, Maki M, Korponay-Szabo I (2005). "Small-bowel mucosal tranglutaminase 2-specific IgA deposits in coeliac disease without villous atrophy: A Prospective and radmonized clinical study". Scand J Gastroenterology 40: 564–572. doi:. PMID 16036509.
- ^ Salmi T, Collin P, Korponay-Szabó I, Laurila K, Partanen J, Huhtala H, Király R, Lorand L, Reunala T, Mäki M, Kaukinen K (2006). "Endomysial antibody-negative coeliac disease: clinical characteristics and intestinal autoantibody deposits". Gut 55 (12): 1746–53. doi:. PMID 16571636. http://gut.bmj.com/cgi/content/full/55/12/1746.
- ^ Londei M, Ciacci C, Ricciardelli I, Vacca L, Quaratino S, and Maiuri L. (2005). "Gliadin as a stimulator of innate responses in celiac disease". Mol Immunol 42 (8): 913–918. doi:. PMID 15829281.
- ^ Zanoni G, Navone R, Lunardi C, Tridente G, Bason C, Sivori S, Beri R, Dolcino M, Valletta E, Corrocher R, Puccetti A (2006). "In celiac disease, a subset of autoantibodies against transglutaminase binds toll-like receptor 4 and induces activation of monocytes". PLoS Med 3 (9): e358. doi:. PMID 16984219. PMC: 1569884. http://medicine.plosjournals.org/perlserv/?request=get-document&doi=10.1371/journal.pmed.0030358.
- ^ Salim A, Phillips A, Farthing M (1990). "Pathogenesis of gut virus infection". Baillieres Clin Gastroenterol 4 (3): 593–607. doi:. PMID 1962725.
- ^ Stene L, Honeyman M, Hoffenberg E, Haas J, Sokol R, Emery L, Taki I, Norris J, Erlich H, Eisenbarth G, Rewers M (2006). "Rotavirus infection frequency and risk of celiac disease autoimmunity in early childhood: a longitudinal study". Am J Gastroenterol 101 (10): 2333–40. doi:. PMID 17032199.
- ^ Kagnoff M, Paterson Y, Kumar P, Kasarda D, Carbone F, Unsworth D, Austin R (1987). "Evidence for the role of a human intestinal adenovirus in the pathogenesis of coeliac disease" (PDF). Gut 28 (8): 995–1001. doi:. PMID 2822550. PMC: 1433141. http://gut.bmj.com/cgi/reprint/28/8/995.
- ^ Suman S, Williams E, Thomas P, Surgenor S, Snook J (2003). "Is the risk of adult coeliac disease causally related to cigarette exposure?". Eur J Gastroenterol Hepatol 15 (9): 995–1000. doi:. PMID 12923372.
- ^ Norris JM, Barriga K, Hoffenberg EJ, Taki I, Miao D, Haas JE, Emery LM, Sokol RJ, Erlich HA, Eisenbarth GS, Rewers M. (2005). "Risk of celiac disease autoimmunity and timing of gluten introduction in the diet of infants at increased risk of disease". JAMA 293 (19): 2343–2351. doi:. PMID 15900004. http://jama.ama-assn.org/cgi/content/full/293/19/2343.
- ^ Poole J, Barriga K, Leung D, Hoffman M, Eisenbarth G, Rewers M, Norris J (2006). "Timing of initial exposure to cereal grains and the risk of wheat allergy". Pediatrics 117 (6): 2175–82. doi:. PMID 16740862. http://pediatrics.aappublications.org/cgi/content/full/117/6/2175.
- ^ Akobeng A, Ramanan A, Buchan I, Heller R (2006). "Effect of breast feeding on risk of coeliac disease: a systematic review and meta-analysis of observational studies". Arch Dis Child 91 (1): 39–43. doi:. PMID 16287899. http://adc.bmj.com/cgi/content/full/91/1/39.
- ^ Treem W (2004). "Emerging concepts in celiac disease". Curr Opin Pediatr 16 (5): 552–9. doi:. PMID 15367850.
- ^ a b Akobeng AK, Thomas AG (June 2008). "Systematic review: tolerable amount of gluten for people with coeliac disease". Aliment. Pharmacol. Ther. 27 (11): 1044–52. doi:. PMID 18315587.
- ^ "Questions and Answers on the Gluten-Free Labeling Proposed Rule". US FDA. 2007-01-23. http://www.cfsan.fda.gov/~dms/glutqa.html. Retrieved on 2008-06-26.
- ^ "Current Official Standards". FAO/WHO. http://www.codexalimentarius.net/web/standard_list.jsp. Retrieved on 2008-06-26.
- ^ a b Troncone R, Ivarsson A, Szajewska H, Mearin ML (June 2008). "Review article: future research on coeliac disease - a position report from the European multistakeholder platform on coeliac disease (CDEUSSA)". Aliment. Pharmacol. Ther. 27 (11): 1030–43. doi:. PMID 18315588. http://www.blackwell-synergy.com/doi/abs/10.1111/j.1365-2036.2008.03669.x.
- ^ Lee AR, Ng DL, Zivin J, Green PH (October 2007). "Economic burden of a gluten-free diet". J Hum Nutr Diet 20 (5): 423–30. doi:. PMID 17845376.
- ^ Häuser W, Stallmach A, Caspary WF, Stein J (March 2007). "Predictors of reduced health-related quality of life in adults with coeliac disease". Aliment. Pharmacol. Ther. 25 (5): 569–78. PMID 17305757.
- ^ Goddard CJ, Gillett HR (November 2006). "Complications of coeliac disease: are all patients at risk?". Postgrad Med J 82 (973): 705–12. doi:. PMID 17099088. http://pmj.bmj.com/cgi/content/full/82/973/705.
- ^ Häuser W, Gold J, Stein J, Caspary WF, Stallmach A (July 2006). "Health-related quality of life in adult coeliac disease in Germany: results of a national survey". Eur J Gastroenterol Hepatol 18 (7): 747–54. doi:. PMID 16772832.
- ^ Siegel M, Bethune M, Gass J, Ehren J, Xia J, Johannsen A, Stuge T, Gray G, Lee P, Khosla C (2006). "Rational design of combination enzyme therapy for celiac sprue". Chem Biol 13 (6): 649–58. doi:. PMID 16793522. http://www.chembiol.com/content/article/fulltext?uid=PIIS1074552106001499.
- ^ Fasano A, Not T, Wang W, Uzzau S, Berti I, Tommasini A, Goldblum S (2000). "Zonulin, a newly discovered modulator of intestinal permeability, and its expression in coeliac disease". Lancet 355 (9214): 1518–9. doi:. PMID 10801176.
- ^ National Institute for Health and Clinical Excellence. Clinical guideline 53: Chronic fatigue syndrome/myalgic encephalomyelitis. London, 2007.
- ^ National Institute for Health and Clinical Excellence. Clinical guideline 61: Irritable bowel syndrome. London, 2008.
- ^ Corazza G, Valentini R, Andreani M, D'Anchino M, Leva M, Ginaldi L, De Feudis L, Quaglino D, Gasbarrini G (1995). "Subclinical coeliac disease is a frequent cause of iron-deficiency anaemia". Scand J Gastroenterol 30 (2): 153–6. doi:. PMID 7732338.
- ^ Ransford R, Hayes M, Palmer M, Hall M (2002). "A controlled, prospective screening study of celiac disease presenting as iron deficiency anemia". J Clin Gastroenterol 35 (3): 228–33. doi:. PMID 12192198.
- ^ Sjöberg K, Carlsson A (2004). "Screening for celiac disease can be justified in high-risk groups" (in Swedish). Lakartidningen 101 (48): 3912, 3915–6, 3918–9. PMID 15631226.
- ^ Catassi C, Rätsch I, Gandolfi L, Pratesi R, Fabiani E, El Asmar R, Frijia M, Bearzi I, Vizzoni L (1999). "Why is coeliac disease endemic in the people of the Sahara?". Lancet 354 (9179): 647–8. doi:. PMID 10466670.
- ^ Zipser R, Farid M, Baisch D, Patel B, Patel D (2005). "Physician awareness of celiac disease: a need for further education". J Gen Intern Med 20 (7): 644–6. doi:10.1111/j.1525-1497.2005.0107.x (inactive 2008-11-08). PMID 16050861. PMC: 1490146. http://www.blackwell-synergy.com/doi/full/10.1111/j.1525-1497.2005.0107.x.
- ^ Fasano A, Berti I, Gerarduzzi T, Not T, Colletti R, Drago S, Elitsur Y, Green P, Guandalini S, Hill I, Pietzak M, Ventura A, Thorpe M, Kryszak D, Fornaroli F, Wasserman S, Murray J, Horvath K (2003). "Prevalence of celiac disease in at-risk and not-at-risk groups in the United States: a large multicenter study". Archives of Internal Medicine 163 (3): 286–92. doi:. PMID 12578508. http://archinte.ama-assn.org/cgi/content/full/163/3/286.
- ^ a b Barker JM, Liu E (2008). "Celiac disease: pathophysiology, clinical manifestations, and associated autoimmune conditions". Adv Pediatr 55: 349–65. doi:. PMID 19048738.
- ^ Leeds JS, Hopper AD, Sanders DS (2008). "Coeliac disease". Br Med Bull 88 (1): 157–70. doi:.
- ^ Jax Peter Lowell, The Gluten-Free Bible, p. 279.
- ^ Scott Adams (2 August 2002). "Bishops in Italy Approve a German-made Low Gluten Eucharistic Host". Celiac.com. http://www.celiac.com/st_prod.html?p_prodid=696.
- ^ Associated Press (8 December 2004). "Girl with digestive disease denied Communion". MSNBC (Microsoft). http://www.msnbc.msn.com/id/5762478/. Retrieved on 2006-05-30.
- ^ Ratzinger, Joseph (24 July 2003). Prot. 89/78-174 98. Congregation for the Doctrine of the Faith. Full text at: "The Use of Mustum and Low-Gluten Hosts at Mass". BCL Newsletter. United States Conference of Catholic Bishops. November 2003. http://www.usccb.org/liturgy/innews/1103.shtml. Retrieved on 2007-03-07.
- ^ McNamara, Father Edward (2004-09-15). "Liturgy: Gluten-free Hosts". Catholic Online. http://www.catholic.org/featured/headline.php?ID=1340. Retrieved on 2007-06-17.
- ^ Rabbi Avraham Juravel. "Gluten Intolerance, Celiac, Allergies And Pesach". Orthodox Union. http://oukosher.org/index.php/articles/single/gluten_intolerance_celiac_allergies_and_pesach/. Retrieved on 2006-09-03.
- ^ a b Holmes, Geoff (2006). "History of coeliac disease". Coeliac UK. http://www.coeliac.co.uk/coeliac_disease/68.asp. Retrieved on 2007-03-20.
- ^ Gee, SJ (1888). "On the coeliac affection". St Bartholomew's Hospital Report 24: 17–20. http://web2.bium.univ-paris5.fr/livanc/?cote=epo0466&p=1&do=page.
- ^ Herter, CA (1908). On infantilism from chronic intestinal infection; characterized by the overgrowth and persistence of flora in the nursing period. New York: Macmillan & Co. as cited by WhoNamedIt
- ^ Ole Daniel Enersen. "Christian Archibald Herter". Who Named It?. http://www.whonamedit.com/doctor.cfm/1490.html. Retrieved on 2007-03-20.
- ^ Haas SV (1924). "The value of the banana in the treatment of coeliac disease". Am J Dis Child 24: 421–37.
- ^ van Berge-Henegouwen G, Mulder C (1993). "Pioneer in the gluten free diet: Willem-Karel Dicke 1905–1962, over 50 years of gluten free diet" (PDF). Gut 34 (11): 1473–5. doi:. PMID 8244125. PMC: 1374403. http://gut.bmj.com/cgi/reprint/34/11/1473.
- ^ Dicke WK. Coeliakie: een onderzoek naar de nadelige invloed van sommige graansoorten op de lijder aan coeliakie [PhD thesis]. Utrecht, the Netherlands: University of Utrecht, 1950.
- ^ Anderson C, French J, Sammons H, Frazer A, Gerrard J, Smellie J (1952). "Coeliac disease; gastrointestinal studies and the effect of dietary wheat flour". Lancet 1 (17): 836–42. doi:. PMID 14918439.
- ^ Paulley JW (1954). "Observation on the aetiology of idiopathic steatorrhoea; jejunal and lymph-node biopsies". Br Med J 4900: 1318–21. PMID 13209109.
- ^ Shiner M (1956). "Duodenal biopsy". Lancet 270 (6906): 17–9. doi:. PMID 13279152.
- ^ Macdonald W, Dobbins W, Rubin C (1965). "Studies of the familial nature of celiac sprue using biopsy of the small intestine". N Engl J Med 272: 448–56. PMID 14242522.
[edit] External links
| Wikibooks Cookbook has a recipe/module on |
- Coeliac disease at the Open Directory Project
- Celiac Disease Awareness Campaign from the National Institutes of Health
- Celiac Disease Foundation - U.S.
- Coeliac UK - leading UK charity
- Celiac Disease practice guideline from the World Gastroenterology Organisation (WGO)
- Celiac Sprue Association - U.S.
- Outcomes of 2004 consensus development conference, U.S. National Institutes of Health
- National Digestive Diseases Clearinghouse - page on coeliac disease
- National Foundation for Celiac Awareness - U.S.
- University of Maryland Center for Celiac Research - U.S.
|
||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||