Serotonin
From Wikipedia, the free encyclopedia
| Serotonin | |
|---|---|
| IUPAC name |
|
| Identifiers | |
| CAS number | 50-67-9 |
| PubChem | |
| MeSH | |
| SMILES |
|
| InChI |
|
| ChemSpider ID | |
| Properties | |
| Molecular formula | C10H12N2O |
| Molar mass | 176.215 |
| Except where noted otherwise, data are given for materials in their standard state (at 25 °C, 100 kPa) Infobox references |
|
Serotonin (pronounced /ˌsɛrəˈtoʊnən/) (5-hydroxytryptamine, or 5-HT) is a monoamine neurotransmitter synthesized in serotonergic neurons in the central nervous system (CNS) and enterochromaffin cells in the gastrointestinal tract of animals including humans. Serotonin is also found in many mushrooms and plants, including fruits and vegetables.
Contents |
[edit] Function

In the central nervous system, serotonin plays an important role as a neurotransmitter in the modulation of anger, aggression, body temperature, mood, sleep, human sexuality, appetite, and metabolism, as well as stimulating vomiting.[1]
Serotonin has broad activities in the brain, and genetic variation in serotonin receptors and the serotonin transporter, which facilitates reuptake of serotonin into presynapses, have been implicated in neurological diseases. Drugs targeting serotonin-induced pathways are being used in the treatment of many psychiatric disorders, and one focus of clinical research is the influence of genetics on serotonin action and metabolism in psychiatric settings. Such studies have revealed that the variation in the promoter region of the serotonin transporter protein accounts for nearly 10% of total variance in anxiety-related personality,[2] and the effect of this gene on depression was found to interact with the environment.[3]
Levels of serotonin in the brain show association with aggression [4], and a mutation in the gene which codes for the 5-HT2A receptor may double the risk of suicide for those with that genotype.[5]
Using the ultimatum game as model, it was shown that people whose serotonin levels have been artificially lowered will reject unfair offers more often than players with normal serotonin levels.[6]
In addition, serotonin is also a peripheral signal mediator. It is found extensively in the human gastrointestinal tract as about 80-90% of the body's total serotonin is found in the enterochromaffin cells in the gut.[7][8] In the blood, the major storage site is platelets, which collect serotonin for use in mediating post-injury vasoconstriction.[9]
Recent research suggests that serotonin plays an important role in liver regeneration and acts as a mitogen (induces cell division) throughout the body. Recent research also suggests that intestinal serotonin may inhibit bone formation.[10][11]
[edit] Serotonin and SIDS
Defective signalling of serotonin in the brain may be the root cause of sudden infant death syndrome (SIDS), Italian researchers have found. Scientists from the European Molecular Biology Laboratory in Monterotondo, Italy,[12] genetically modified lab mice to produce low levels of the brain signaling protein serotonin. The results showed the mice suffered drops in heart rate and other symptoms of SIDS, and many of the animals died at an early age.
Researchers now believe that low levels of serotonin in the animals' brainstems, which control heartbeat and breathing, may have caused sudden death, researchers said in the July 4, 2008 issue of Science.[13]
[edit] Anatomy

[edit] Gross anatomy
The neurons of the raphe nuclei are the principal source of 5-HT release in the brain.[14] The raphe nuclei are neurons grouped into about nine pairs and distributed along the entire length of the brainstem, centered around the reticular formation.[15]
Axons from the neurons of the raphe nuclei form a neurotransmitter system, reaching large areas of the brain. Axons of neurons in the caudal raphe nuclei terminate in the following locations:
On the other hand, axons of neurons in the rostral raphe nuclei terminate in e.g.:
Thus, activation of this serotonin system has effects on large areas of the brain.
[edit] Microanatomy
Serotonin is released from serotonergic varicosities (swellings) into the extra neuronal space, but not from synaptic terminal boutons as other neurotransmitters.[citation needed] Serotonin diffuses over a relatively wide gap (>20µm) to activate 5-HT receptors located on the dendrites, cell bodies and presynaptic terminals of adjacent neurons.
[edit] Receptors
5-HT receptors are the receptors for serotonin. They are located on the cell membrane of nerve cells and other cell types in animals and mediate the effects of serotonin as the endogenous ligand and of a broad range of pharmaceutical and hallucinogenic drugs. With the exception of the 5-HT3 receptor, a ligand gated ion channel, all other 5-HT receptors are G protein coupled seven transmembrane (or heptahelical) receptors that activate an intracellular second messenger cascade.[citation needed]
[edit] Termination
Serotonergic action is terminated primarily via uptake of 5-HT from the synapse. This is through the specific monoamine transporter for 5-HT, SERT, on the presynaptic neuron. Various agents can inhibit 5-HT reuptake including MDMA (ecstasy), amphetamine, cocaine, dextromethorphan (an antitussive), tricyclic antidepressants (TCAs) and selective serotonin reuptake inhibitors (SSRIs).
Interestingly, a 2006 study conducted by the University of Washington suggested that a newly discovered monoamine transporter, known as PMAT, may account for 'a significant percentage of 5-HT clearance.[16]' Contrasting with the high-affinity SERT, the PMAT has been identified as a low affinity transporter with an apparent Km of 114 micromoles/L for serotonin; approximately 230 times higher than that of SERT. However, the PMAT, despite its relatively low serotonergic affinity, has a considerably higher transport capacity than SERT,
"..resulting in roughly comparable uptake efficiencies to SERT in heterologous expression systems."
The study also suggests that some SSRIs, such as fluoxetine and sertraline, inhibit PMAT but at IC50 values which surpass therapeutic plasma concentrations by up to four magnitudes of ten; ergo, SSRI monotherapy is ineffective in PMAT inhibition. At present, there are no known pharmaceuticals which would appreciably inhibit PMAT at normal therapeutic doses. The PMAT also suggestively transports dopamine and norepinephrine albeit at Km values even higher than that of 5-HT (330–15,000 micromoles/L).
[edit] Endothelial cell function and Serotonin
5-hydroxytryptamine evokes endothelial nitric oxide synthase activation and stimulates phosphorylation of p44/p42 mitogen-activated protein kinase activation in bovine aortic endothelial cell cultures.[17]
[edit] Biosynthesis
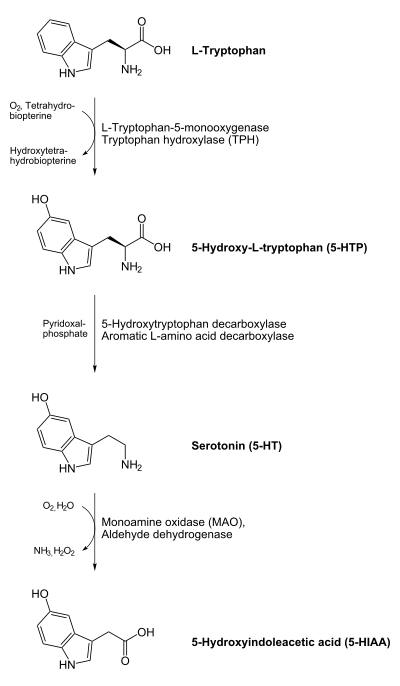
In animals including humans, serotonin is synthesized from the amino acid L-tryptophan by a short metabolic pathway consisting of two enzymes: tryptophan hydroxylase (TPH) and amino acid decarboxylase (DDC). The TPH-mediated reaction is the rate-limiting step in the pathway. TPH has been shown to exist in two forms: TPH1, found in several tissues, and TPH2, which is a brain-specific isoform. There is evidence that genetic polymorphisms in both these subtypes influence susceptibility to anxiety and depression in humans. There is also evidence that ovarian hormones can affect the expression of TPH in various species, suggesting a possible mechanism for postpartum depression and premenstrual stress syndrome.[citation needed] Serotonin biosynthesis in plants likewise begins with L-tryptophan, which is however first decarboxylated by tryptophan decarboxylase to give tryptamine, which is then hydroxylated by the cytochrome P450 monooxygenase, tryptamine 5-hydroxylase, yielding serotonin.[18]
Serotonin taken orally does not pass into the serotonergic pathways of the central nervous system because it does not cross the blood-brain barrier. However, tryptophan and its metabolite 5-hydroxytryptophan (5-HTP), from which serotonin is synthesized, can and do cross the blood-brain barrier. These agents are available as dietary supplements and may be effective serotonergic agents.
One product of serotonin breakdown is 5-Hydroxyindoleacetic acid (5 HIAA), which is excreted in the urine. Serotonin and 5 HIAA are sometimes produced in excess amounts by certain tumors or cancers, and levels of these substances may be measured in the urine to test for these tumors.
[edit] Drugs targeting the 5-HT system
Several classes of drugs target the 5-HT system including some antidepressants, antipsychotics, anxiolytics, antiemetics, and antimigraine drugs as well as the psychedelic drugs and empathogens.
[edit] Psychedelic drugs
The psychedelic drugs psilocin/psilocybin, DMT, mescaline, and LSD mimic the action of serotonin primarily at 5-HT2A receptor. The empathogen MDMA (ecstasy) releases serotonin from synaptic vesicles of neurons.
[edit] Antidepressants
The MAOIs prevent the breakdown of monoamine neurotransmitters (including serotonin), and therefore increase concentrations of the neurotransmitter in the brain. MAOI therapy is associated with many adverse drug reactions, and patients are at risk of hypertensive emergency triggered by foods with high tyramine content and certain drugs.
Some drugs inhibit the re-uptake of serotonin, making it stay in the synapse longer. The tricyclic antidepressants (TCAs) inhibit the re-uptake of both serotonin and norepinephrine. The newer selective serotonin re-uptake inhibitors (SSRIs) have fewer side-effects and fewer interactions with other drugs.
Certain SSRI medications have been shown to lower serotonin levels below the baseline after chronic use; despite initial increases in serotonin. This attributes to the observation that the benefit of SSRI's may decrease in select patients after a long-term treatment. A switch in medication will usually resolve this issue; up to 70% of the time. [19] However, the novel antidepressant Tianeptine, selective serotonin reuptake enhancer, has mood elevating effects. This has given evidence to the theory that serotonin is most likely used to regulate the extent or intensity of moods.
[edit] Antiemetics
5-HT3 antagonists such as ondansetron, granisetron, and tropisetron are important antiemetic agents. They are particularly important in treating the nausea and vomiting that occur during anticancer chemotherapy using cytotoxic drugs. Another application is in the treatment of post-operative nausea and vomiting. Applications to the treatment of depression and other mental and psychological conditions have also been investigated with some positive results.
[edit] Pathology
If neurons that make serotonin — serotonergic neurons — are abnormal in infants, there is a risk of sudden infant death syndrome (SIDS).[20][21] Low levels of serotonin may also be associated with intense spiritual experiences.[22]
Recent research conducted at Rockefeller University shows that both in patients who suffer from depression and in mice that model the disorder, levels of the p11 protein are decreased. This protein is related to serotonin transmission within the brain.[23]
Obsessive-compulsive disorder (OCD) can be a debilitating disorder with the following two anxiety-related essential features: obsessions (undesirable, recurrent, disturbing thoughts) and compulsions (repetitive or ritualized behaviors). SSRIs, and other medicines which alter serotonin levels, have been approved to be used to treat symptoms of OCD.
[edit] Serotonin syndrome
Extremely high levels of serotonin can have toxic and potentially fatal effects, causing a condition known as serotonin syndrome. In practice, such toxic levels are essentially impossible to reach through an overdose of a single anti-depressant drug, but require a combination of serotonergic agents, such as an SSRI with an MAOI.[24] The intensity of the symptoms of serotonin syndrome vary over a wide spectrum, and the milder forms are seen even at non-toxic levels.[25][citation needed]
[edit] Chronic diseases resulting from serotonin 5-HT2B overstimulation
In blood, serotonin stored in platelets is active wherever platelets bind, as a vasoconstrictor to stop bleeding, and also as a fibrocyte mitotic, to aid healing. Because of these effects, overdoses of serotonin, or serotonin agonist drugs, may cause acute or chronic pulmonary hypertension from pulmonary vasoconstriction, or else syndromes of retroperitoneal fibrosis or cardiac valve fibrosis (endocardial fibrosis) from overstimulation of serotonic growth receptors on fibrocytes.[citation needed]
Serotonin itself may cause a syndrome of cardiac fibrosis when it is eaten in large quantities in the diet (the Matoki banana of East Africa) or when it is over-secreted by certain mid-gut carcinoid tumors.[citation needed] The valvular fibrosis in such cases is typically on the right side of the heart, since excess serotonin in the serum outside platelets is metabolized in the lungs, and does not reach the left circulation.[citation needed]
Serotonergic agonist drugs in overdose in experimental animals not only cause acute (and sometimes fatal) pulmonary hypertension, but there is epidemiologic evidence that chronic use of certain of these drugs produce a chronic pulmonary hypertensive syndrome in humans.[citation needed] Some serotonergic agonist drugs also cause fibrosis anywhere in the body, particularly the syndrome of retroperitoneal fibrosis, as well as cardiac valve fibrosis.[26]
In the past, three groups of serotonergic drugs have been epidemiolgically linked with these syndromes. They are the serotonergic vasoconstrictive anti-migraine drugs (ergotamine and methysergide),[26] the serotonergic appetite suppressant drugs (fenfluramine, chlorphentermine, and aminorex), and certain anti-parkinsonian dopaminergic agonists, which also stimulate serotonergic 5-HT2B receptors. These include pergolide and cabergoline, but not the more dopamine-specific lisuride.[27] As with fenfluramine, some of these drugs have been withdrawn from the market after groups taking them showed a statistical increase of one or more of the side effects described. An example is pergolide. The drug was in decreasing use since reported in 2003 to be associated with cardiac fibrosis.[28] Two independent studies published in the New England Journal of Medicine in January 2007, implicated pergolide along with cabergoline in causing valvular heart disease.[29][30] As a result of this, the FDA removed pergolide from the U.S. market in March, 2007.[31] (Since cabergoline is not approved in the U.S. for Parkinson's Disease, but for hyperprolactinemia, the drug remains on the market. Treatment for hyperprolactinemia requires lower doses than that for Parkinson's Disease, diminishing the risk of valvular heart disease).[32]
Because neither the amino acid L-tryptophan nor the SSRI-class antidepressants raise blood serotonin levels[citation needed], they are not under suspicion to cause the syndromes described. However, since 5-hydroxytryptophan (5-HTP) does raise blood serotonin levels, it is under some of the same scrutiny as actively serotonergic drugs.[citation needed]
[edit] In unicellular organisms
Serotonin is used by a variety of single-cell organisms for various purposes. Selective serotonin re-uptake inhibitors (SSRIs) have been found to be toxic to algae.[33] The gastrointestinal parasite Entamoeba histolytica secretes serotonin, causing a sustained secretory diarrhea in some patients.[34][35] Patients infected with Entamoeba histolytica have been found to have highly elevated serum serotonin levels which returned to normal following resolution of the infection.[36] Entamoeba histolytica also responds to the presence of serotonin by becoming more virulent.[37]
[edit] In plants
Serotonin is found in mushrooms and plants, including fruits and vegetables. The highest values of 25–400 mg/kg have been found in nuts of the walnut (Juglans) and hickory (Carya) genuses. Serotonin concentrations of 3–30 mg/kg have been found in plantain, pineapple, banana, kiwifruit, plums, and tomatoes. Moderate levels from 0.1–3 mg/kg have been found in a wide range of tested vegetables.[38] Serotonin is one compound of the poison contained in stinging nettles (Urtica dioica). It should be noted that serotonin, unlike its precursors 5-HTP and tryptophan, does not cross the blood–brain barrier, which means that ingesting serotonin in the diet has no effect on brain serotonin levels. Several plants contain serotonin together with a family of related tryptamines that are methylated at the amino (NH2) and hydroxy (OH) groups, are N-oxides, or miss the OH group. Examples are plants from the Anadenanthera genus that are used in the hallucinogenic yopo snuff.
[edit] In animals
Serotonin as a neurotransmitter is found in many animals, including insects. Several toad venoms, as well as that of the Brazilian wandering spider and stingray, contain serotonin and related tryptamines. It has also been identified as the trigger for swarm behaviour in locusts[39].
[edit] History
Isolated and named in 1948 by Maurice M. Rapport, Arda Green, and Irvine Page of the Cleveland Clinic,[40] the name serotonin is something of a misnomer and reflects the circumstances of the compound's discovery. It was initially identified as a vasoconstrictor substance in blood serum – hence serotonin, a serum agent affecting vascular tone. This agent was later chemically identified as 5-hydroxytryptamine (5-HT) by Rapport, and, as the broad range of physiological roles were elucidated, 5-HT became the preferred name in the pharmacological field.
[edit] Increasing serotonin levels
| This article or section appears to contradict itself. Please help fix this problem. |
Serotonin levels can not be increased by diet or supplements of tryptophan alone. For example, increasing foods rich in tryptophan (eg, meats, proteins) does not increase serotonin levels, due to competition with other amino acids.[41] What is required to increase serotonin production is an increase in the ratio of tryptophan to phenylalanine and leucine. Fruits with a good ratio include dates, papaya and banana. Foods with a lower ratio inhibit the production of serotonin. These include whole wheat and rye bread[42] Much research has indicated that vigorous aerobic exercise improves mood, believed to be facilitated by an increase in serotonin levels.[43] Research also suggests that eating a diet rich in whole grain carbohydrates and low in protein will increase serotonin by secreting insulin, which helps in amino acid competition.[41] However, increasing insulin for a long period of time can sometimes onset insulin resistance, which is related to obesity, type 2 diabetes, and lower serotonin levels. It is also believed that muscles use many of the amino acids except tryptophan, allowing men to have more serotonin than women.[44] Bright light therapy is another popular method which prevents the conversion of serotonin to melatonin.[45] A similar effect is obtained by spending more time in natural sunlight. Recently, acupuncture has been shown to stimulate the release of serotonin in lab animals.[46]
[edit] References
- ^ Dolasetron
- ^ Lesch, K.; Bengel, D.; Heils, A.; Sabol, S. Z.; Greenberg, B. D.; Petri, S.; Clemens, R.; Müller, J. B.; Hamer, D. H.; Murphy, D. L. (1996). "Association of Anxiety-Related Traits with a Polymorphism in the Serotonin Transporter Gene Regulatory Region". Science 274: 1527–31. doi:. PMID 8929413.
- ^ Caspi, A.; Sugden, K.; Moffitt, T. E.; Taylor, A.; Craig, I. W.; Harrington, W.; McClay, J.; Mill, J.; Martin, J.; Braithwaite, A.; Poulton, R. (2003). "Influence of life stress on depression: moderation by a polymorphism in the 5-HTT gene". Science 301: 386–89. doi:. PMID 12869766.
- ^ Caspi N, Modai I, Barak P, Waisbourd A, Zbarsky H, Hirschmann S, Ritsner M. (2001 Mar). "Pindolol augmentation in aggressive schizophrenic patients: a double-blind crossover randomized study". Int Clin Psychopharmacol. 16(2): 111-5. PMID 11236069.
- ^ Basky, Greg (May 2000). "Suicide linked to serotonin gene". CMAJ 162 (9). http://www.cmaj.ca/cgi/content/full/162/9/1343-a.
- ^ Crockett, M. J.; Clark, L.; Tabibnia, G.; Lieberman, M. D.; Robbins, T. W. (June 2008). "Serotonin modulates behavioral reactions to unfairness". Science (journal) 320 (5884): 1739. doi:. PMID 18535210.
- ^ Indiana State University
- ^ [1] Serotonin tests info, Accessed May 6, 2008
- ^ Essentials of Human Anatomy & Physiology, Eighth Edition, p. 336
- ^ http://www.sciencedaily.com/releases/2008/11/081126122209.htm
- ^ Cell 135, 825–837, November 28, 2008
- ^ Audero, Enrica; Coppi, Elisabetta; Mlinar, Boris; Rossetti, Tiziana; Caprioli, Antonio; Al Banchaabouchi, Mumna; Corradetti, Renato; Gross, Cornelius (2008). "Sporadic Autonomic Dysregulation and Death Associated with Excessive Serotonin Autoinhibition". Science 321: 130–133. doi:. PMID 18599790.
- ^ Lesurtel, M. et al. (2006). "Platelet-derived serotonin mediates liver regeneration". Science 312 (5770): 104–7. doi:. PMID 16601191.
- ^ Frazer, A.; and Hensler, J. G. (1999). "Understanding the neuroanatomical organization of serotonergic cells in the brain provides insight into the functions of this neurotransmitter". in Siegel, G. J.. Basic Neurochemistry. Agranoff, Bernard W.; Fisher, Stephen K.; Albers, R. Wayne; Uhler, Michael D. (Sixth ed.). Lippincott Williams and Wilkins. ISBN 0-397-51820-X. http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Search&db=books&doptcmdl=GenBookHL&term=raphe+AND+serotonin+release+AND+bnchm%5Bbook%5D+AND+160428%5Buid%5D&rid=bnchm.section.946#949. "In 1964, Dahlstrom and Fuxe (discussed in [2]), using the Falck-Hillarp technique of histofluorescence, observed that the majority of serotonergic soma are found in cell body groups, which previously had been designated as the raphe nuclei."
- ^ |The Raphe nuclei group of neurons are located along the brain stem from the labels 'Mid Brain' to 'Oblongata', centered on the pons. (See relevant image.)
- ^ Mingyan Zhou, Karen Engel,1 and Joanne Wang* (2007). "Evidence for Significant Contribution of a Newly Identified Monoamine Transporter (PMAT) to Serotonin Uptake in the Human Brain". in ?. Biochemical Pharmacology. ? (Vol 73 Issue 1 ed.). Elsevier Inc.. doi:. ISBN ?. http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=1828907.
- ^ McDuffie, J. E. (March 2000). "-hydroxytryptamine stimulates phosphorylation of p44/p42 mitogen-activated protein kinase activation in bovine aortic endothelial cell cultures". J. Cardiovasc. Pharmacol. 35 (3). http://www.cardiovascularpharm.com/pt/re/jcardiopharm/abstract.00005344-200003000-00008.htm;jsessionid=LLhXJN6PTXBlSKWpG3mTNChLl7C4wQZFQF2yyFWnlQLcdph7zkQq!-1990489359!181195628!8091!-1.
- ^ Schröder P, Abele C, Gohr P, Stuhlfauth-Roisch U, Grosse W. (1999). "Latest on enzymology of serotonin biosynthesis in walnut seeds". Adv Exp Med Biol. 467: 637-644. PMID 10721112.
- ^ Benmansour, Saloua et al., Effects of Chronic Antidepressant Treatments on Serotonin Transporter Function, Density, and mRNA Level. Journal of Neuroscience, 1999, 19(23):10494-10501. http://www.jneurosci.org/cgi/content/full/19/23/10494
- ^ Paterson, D. S.; Trachtenberg, F. L.; Thompson, E.G.; et al. (November 2006). "Multiple serotonergic brainstem abnormalities in sudden infant death syndrome". JAMA 296 (17): 2124–32. doi:. PMID 17077377.
- ^ Sciencedaily Report Anger and Aggression in Women: Blame It On Genetics
- ^ Farde, Lars; and Borg, Jacqueline; Section of psychiatry at Karolinska Institutet in Stockholm, Sweden 2003, the study and a vulgarized article
- ^ Svenningsson, P. et al. (2006). "Alterations in 5-HT1B receptor function by p11 in depression-like states". Science 311 (5757): 77–80. doi:. PMID 16400147.
- ^ Isbister, G. K.; Bowe, S. J.; Dawson, A.; Whyte, I. M. (2004). "Relative toxicity of selective serotonin reuptake inhibitors (SSRIs) in overdose". J. Toxicol. Clin. Toxicol. 42 (3): 277–85. PMID 15362595.
- ^ Dunkley, E. J.; Isbister, G. K.; Sibbritt, D.; Dawson, A. H.; Whyte, I. M. (September 2003). "The Hunter Serotonin Toxicity Criteria: simple and accurate diagnostic decision rules for serotonin toxicity". QJM 96 (9): 635–42. PMID 12925718. http://qjmed.oxfordjournals.org/cgi/pmidlookup?view=long&pmid=12925718.
- ^ a b Baskin, Steven I. (1991). Principles of Cardiac Toxicology. CRC Press. ISBN 0849388090. http://books.google.com/books?id=AW7M6jBixj4C&pg=PA626&lpg=PA626&dq=ergotamine+endocardial+fibrosis&source=web&ots=BrjNzsIc-N&sig=X-WM0TaoW6I5nkx6xiTsxJYvLHo..
- ^ Jähnichen, S.; Horowski, R.; Pertz, H. "Pergolide and Cabergoline But not Lisuride Exhibit Agonist Efficacy at Serotonin 5-HT2B Receptors".PDF (515 KB) Presentation. Retrieved on 2007-03-30.
- ^ ADRAC (August 2004). "Cardiac valvulopathy with pergolide". Aust Adv Drug React Bull 23 (4). http://www.tga.gov.au/adr/aadrb/aadr0408.htm. Free full text from the Australian Therapeutic Goods Administration
- ^ Schade, Rene; Andersohn, Frank; Suissa, Samy; Haverkamp, Wilhelm; Garbe, Edeltraut (2007-01-04), "Dopamine Agonists and the Risk of Cardiac-Valve Regurgitation", New England Journal of Medicine 356 (1): 29–38, PMID 17202453, http://content.nejm.org/cgi/content/full/356/1/29
- ^ Zanettini, Renzo; Antonini, Angelo; Gatto, Gemma; Gentile, Rosa; Tesei, Silvana; Pezzoli, Gianna (2007-01-04), "Valvular Heart Disease and the Use of Dopamine Agonists for Parkinson's Disease", New England Journal of Medicine 356 (1): 39–46, PMID 17202454, http://content.nejm.org/cgi/content/full/356/1/39
- ^ "Food and Drug Administration Public Health Advisory". 2007-03-29. http://www.fda.gov/cder/drug/advisory/pergolide.htm. Retrieved on 2007-04-27.
- ^ "MedWatch - 2007 Safety Information Alerts. Permax (pergolide) and generic equivalents". U.S. Food and Drug Administration. March 29, 2007. http://www.fda.gov/medwatch/safety/2007/safety07.htm#Pergolide. Retrieved on 2007-03-30.
- ^ Johnson, D. J.; Sanderson, H.; Brain, R. A.; Wilson, C. J.; Solomon, K. R. (2007). "Toxicity and hazard of selective serotonin reuptake inhibitor antidepressants fluoxetine, fluvoxamine, and sertraline to algae". Ecotoxicol. Environ. Saf. 67 (1): 128–39. doi:. PMID 16753215.
- ^ McGowan, K.; Kane, A.; Asarkof, N. et al. (1983). "Entamoeba histolytica causes intestinal secretion: role of serotonin". Science 221 (4612): 762–4. doi:. PMID 6308760.
- ^ McGowan, K.; Guerina, V.; Wicks, J.; Donowitz, M. (1985). "Secretory hormones of Entamoeba histolytica". Ciba Found. Symp. 112: 139–54. PMID 2861068.
- ^ Banu, Naheed et al. (2005). "Neurohumoral alterations and their role in amoebiasis" (PDF). Indian J. Clin Biochem 20 (2): 142–5. doi:. http://medind.nic.in/iaf/t05/i2/iaft05i2p142.pdf.
- ^ Acharya, D. P.; Sen, M. R.; Sen, P. C. (1989). "Effect of exogenous 5-hydroxytryptamine on pathogenicity of Entamoeba histolytica in experimental animals". Indian J. Exp. Biol. 27 (8): 718–20. PMID 2561282.
- ^ Feldman, J. M.; Lee, E. M. (October 1985). "Serotonin content of foods: effect on urinary excretion of 5-hydroxyindoleacetic acid". Am. J. Clin. Nutr. 42 (4): 639–43. PMID 2413754. http://www.ajcn.org/cgi/pmidlookup?view=long&pmid=2413754.
- ^ http://news.bbc.co.uk/2/hi/science/nature/7858996.stm
- ^ Rapport, Maurice M.; Green, Arda A.; Page, Irvine H. (December 1948). "Serum vasoconstrictor (serotonin). IV. Isolation and characterization". J. Biol. Chem. 176 (3): 1243–51. PMID 18100415.
- ^ a b Young, S. N. (November 2007). "How to increase serotonin in the human brain without drugs" (PDF). J. Psychiatry Neurosci. 32 (6): 394–9. PMID 18043762. PMC: 2077351. http://www.cma.ca/multimedia/staticContent/HTML/N0/l2/jpn/vol-32/issue-6/pdf/pg394.pdf. Retrieved on 2008-12-30.
- ^ Nutritional Pharmacology Of Sleep & Depression
- ^ Serotonin and Its Uses
- ^ http://books.google.com/books?id=2PxhgaCThccC&pg=PA49&lpg=PA49&dq=insulin+resistance+and+serotonin&source=web&ots=udW-o42zjY&sig=rtcdA8TTAkuxSxwvrR4TxYcnF30&hl=en&sa=X&oi=book_result&resnum=3&ct=result#PPA64,M1
- ^ Education : Philips
- ^ Acupuncture Stimulates the Release of Serotonin, but Not Dopamine, in the Rat Nucleus Accumbens
[edit] External links
- PsychoTropicalResearch Extensive reviews on serotonergic drugs and Serotonin Syndrome.
- Molecule of the Month: Serotonin at University of Bristol
- Scientific America 60-Second Psych: No Fair! My Serotonin Level Is Low.
- Serotonin Test Interpretation on ClinLab Navigator.
|
|||||
|
|||||