Metformin
From Wikipedia, the free encyclopedia
 |
|
 |
|
|
Metformin
|
|
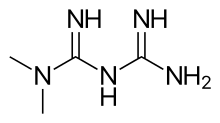
| Systematic (IUPAC) name | |
| N,N-dimethylimidodicarbonimidic diamide | |
| Identifiers | |
| CAS number | |
| ATC code | A10 A10 (with sulfonylureas) A10 (with rosiglitazone) A10 (with pioglitazone) A10 (with sitagliptin) A10 (with vildagliptin) |
| PubChem | |
| DrugBank | |
| Chemical data | |
| Formula | C4H11N5 |
| Mol. mass | 129.164 g/mol 165.63 g/mol (hydrochloride) |
| Synonyms | 1,1-dimethylbiguanide |
| Pharmacokinetic data | |
| Bioavailability | 50 to 60% under fasting conditions |
| Metabolism | None |
| Half life | 6.2 hours |
| Excretion | Active renal tubular excretion by OCT2 |
| Therapeutic considerations | |
| Licence data | |
| Pregnancy cat. | |
| Legal status | |
| Routes | Oral |
Metformin (INN; trade names Glucophage, Riomet, Fortamet, Glumetza, Obimet, Dianben, Diabex, Diaformin, and others) (IPA: /mɛtˈfɔrmɪn/) is an oral anti-diabetic drug from the biguanide class. It is the first-line drug for the treatment of type 2 diabetes, particularly in overweight and obese people and those with normal kidney function,[1][2][3] and evidence suggests it may be the best choice for people with heart failure.[4] It is also used in the treatment of polycystic ovary syndrome.
Metformin is the most popular anti-diabetic drug in the United States and one of the most prescribed drugs in the country overall, with nearly 35 million prescriptions filled in 2006 for generic metformin alone.[5] When prescribed appropriately, metformin causes few adverse effects—the most common is gastrointestinal upset—and, unlike many other anti-diabetic drugs, does not cause hypoglycemia if used alone. It also helps reduce LDL cholesterol and triglyceride levels, and may aid weight loss. As of 2008[update], metformin is one of only two oral anti-diabetics in the World Health Organization Model List of Essential Medicines (the other being glibenclamide).[6]
Contents |
[edit] History
The biguanide class of anti-diabetic drugs, which also includes the withdrawn agents phenformin and buformin, originates from the French lilac (Galega officinalis), a plant known for several centuries to reduce the symptoms of diabetes mellitus.[7]
Metformin was first described in the scientific literature in 1957.[8] It was first marketed in France in 1979, but did not receive approval by the U.S. Food and Drug Administration (FDA) for Type 2 diabetes until 1994.[9] Bristol-Myers Squibb's Glucophage was the first branded formulation of metformin to be marketed in the United States, beginning on March 3, 1995.[10] Generic formulations are now available.
[edit] Indications
The main use for metformin is in the treatment of diabetes mellitus type 2, especially when this accompanies obesity and insulin resistance. Metformin is the only anti-diabetic drug that has been proven to protect against the cardiovascular complications of diabetes.[11] This was first shown in the United Kingdom Prospective Diabetes Study, a large study of overweight patients with diabetes.[12] Unlike the other most-commonly prescribed class of oral diabetes drugs, the sulfonylureas, metformin (taken alone) does not induce hypoglycemia.[13] Hypoglycemia during intense exercise has been documented, but is extremely rare.[14] It also does not cause weight gain, and may indeed produce minor weight loss.[15] Metformin also modestly reduces LDL and triglyceride levels.[16]
It is also being used increasingly in polycystic ovary syndrome (PCOS),[17] non-alcoholic fatty liver disease (NAFLD)[18] and premature puberty,[19] three other diseases that feature insulin resistance; these indications are still[update] considered experimental. Although metformin is not licensed for use in PCOS, the United Kingdom's National Institute for Health and Clinical Excellence recommends that women with PCOS and a body mass index above 25 be given metformin when other therapy has failed to produce results.[20] The benefit of metformin in NAFLD has not been extensively studied and may be only temporary.[21]
It may reduce weight gain in patients taking atypical antipsychotics.[22]
[edit] Contraindications
Metformin is contraindicated in people with any condition that could increase the risk of lactic acidosis, including kidney disorders (creatinine levels over 150 μmol/l,[23] although this is an arbitrary limit), lung disease and liver disease. Heart failure has long been considered a contraindication for metformin use, although a 2007 systematic review showed metformin to be the only anti-diabetic drug not associated with harm in people with heart failure.[4]
It is recommended that metformin be temporarily discontinued before any radiographic study involving iodinated contrast (such as a contrast-enhanced CT scan or angiogram), as contrast dye may temporarily impair kidney function, indirectly leading to lactic acidosis by causing retention of metformin in the body.[24][25] It is recommended that metformin be resumed after two days, assuming kidney function is normal.[24][25]
[edit] Adverse effects
[edit] Lactic acidosis
The most serious potential side effect of metformin is lactic acidosis; this complication is very rare, and seems limited to those with impaired liver or kidney function. Phenformin, another biguanide, was withdrawn because of an increased risk of lactic acidosis (up to 60 cases per million patient-years). However, metformin is safer than phenformin, and the risk of developing lactic acidosis is not increased by the medication so long as it is not prescribed to known high-risk groups.[26]
[edit] Gastrointestinal
The most common adverse effect of metformin is gastrointestinal upset, including diarrhea, cramps, nausea, vomiting and increased flatulence; metformin is more commonly associated with gastrointestinal side effects than most other anti-diabetic drugs.[16] In a clinical trial of 286 subjects, 53.2% of the 141 who were given immediate-release metformin (as opposed to placebo) reported diarrhea, versus 11.7% for placebo, and 25.5% reported nausea/vomiting, versus 8.3% for those on placebo.[27]
Gastrointestinal upset can cause severe discomfort for patients; it is most common when metformin is first administered, or when the dose is increased. The discomfort can often be avoided by beginning at a low dose (1 to 1.7 grams per day) and increasing the dose gradually. Gastrointestinal upset after prolonged, steady use is less common.
Long-term use of metformin has been associated with increased homocysteine levels[28] and malabsorption of vitamin B12.[29][30] Higher doses and prolonged use are associated with increased incidence of B12 deficiency, and some researchers recommend screening or prevention strategies.[31]
[edit] Overdosage
A review of intentional and accidental metformin overdoses reported to Poison control centers over a 5-year period found that serious adverse events were rare, though elderly patients appeared to be at greater risk.[32] Intentional overdoses with up to 63 g of metformin have been reported in the medical literature.[33] The major potentially life-threatening complication of metformin overdose is lactic acidosis. Treatment of metformin overdose is generally supportive, but may include sodium bicarbonate to address acidosis and standard hemodialysis or continuous veno-venous hemofiltration to rapidly remove metformin and correct acidosis.[34][35]
[edit] Mechanism of action
Metformin improves hyperglycemia primarily through its suppression of hepatic glucose production (hepatic gluconeogenesis).[36] The "average" person with type 2 diabetes has three times the normal rate of gluconeogenesis; metformin treatment reduces this by over one third.[37] Metformin activates AMP-activated protein kinase (AMPK), a liver enzyme that plays an important role in insulin signaling, whole body energy balance, and the metabolism of glucose and fats;[38] activation of AMPK is required for metformin's inhibitory effect on the production of glucose by liver cells.[39] Research published in 2008 further elucidated metformin's mechanism of action, showing that activation of AMPK is required for an increase in the expression of SHP, which in turn inhibits the expression of the hepatic gluconeogenic genes PEPCK and Glc-6-Pase.[40] Metformin is frequently used in research along with AICAR as an AMPK agonist. The mechanism by which biguanides increase the activity of AMPK remains uncertain; however, research suggests that metformin increases the amount of cytosolic AMP (as opposed to a change in total AMP or total AMP/ATP).[41]
In addition to suppressing hepatic glucose production, metformin increases insulin sensitivity, enhances peripheral glucose uptake, increases fatty acid oxidation,[42] and decreases absorption of glucose from the gastrointestinal tract. Increased peripheral utilization of glucose may be due to improved insulin binding to insulin receptors.[43] AMPK probably also plays a role, as metformin administration increases AMPK activity in skeletal muscle.[44] AMPK is known to cause GLUT4 translocation, resulting in insulin-independent glucose uptake. Some metabolic actions of metformin do appear to occur by AMPK-independent mechanisms; a recent study found that "the metabolic actions of metformin in the heart muscle can occur independent of changes in AMPK activity and may be mediated by p38 MAPK- and PKC-dependent mechanisms."[45]
Metformin is not metabolized, rather it is primarily excreted in the urine with an elimination half-life of 6.2 hours.[46]
[edit] Interactions
The H2-receptor antagonist cimetidine causes an increase in the plasma concentration of metformin, by reducing clearance of metformin by the kidneys;[47] both metformin and cimetidine are cleared from the body by tubular secretion, and both, particularly the cationic (positively charged) form of cimetidine, may compete for the same transport mechanism.[48] A small double-blind, randomized study found the antibiotic cefalexin to also increase metformin concentrations by a similar mechanism;[49] theoretically, other cationic medications may produce the same effect.[48]
[edit] Formulations
Metformin IR (immediate release) is available in 500 mg, 850 mg, and 1000 mg tablets, all now generic in the US.
Metformin SR (slow release) or XR (extended release) was introduced in 2004, in 500 mg and 750 mg strengths, mainly to counteract the most common gastrointestinal side effects, as well as to increase patient compliance by reducing pill burden. No difference in effectiveness exists between the two preparations.
[edit] Combinations with other drugs
Metformin is often prescribed to type 2 diabetes patients in combination with rosiglitazone. This drug actively reduces insulin resistance, complementing the action of the metformin. In 2002, the two drugs were combined into a single product, Avandamet, marketed by GlaxoSmithKline.[50] In 2005, all current stock of Avandamet was seized by the FDA and removed from the market, after inspections showed the factory where it was produced was violating Good Manufacturing Practices.[51] The drug pair continued to be prescribed separately in the absence of Avandamet, which was available again by the end of that year.
In the United States, metformin is also available in combination with pioglitazone (trade name Actoplus Met), the sulfonylureas glipizide (trade name Metaglip) and glibenclamide (known as glyburide in the United States, trade name Glucovance), the dipeptidyl peptidase-4 inhibitor sitagliptin (trade name Janumet), and the meglitinide repaglinide (PrandiMet). Generic formulations of metformin/glipizide and metformin/glibenclamide are available. A generic formulation of metformin/rosiglitazone from Teva has received tentative approval from the FDA, and is expected to reach the market in early 2012.[52]
[edit] References
- ^ Clinical Guidelines Task Force, International Diabetes Federation (2005). "Glucose control: oral therapy"PDF (100 KB). In: Global Guideline for Type 2 Diabetes. Brussels: International Diabetes Federation, 35–8. Retrieved on November 6, 2007.
- ^ National Institute for Health and Clinical Excellence. Clinical guideline 66: Diabetes - type 2 (update). London, 2008.
- ^ American Diabetes Association (2007). "Standards of medical care in diabetes—2007". Diabetes Care 30 Suppl 1: S4–S41. doi:. PMID 17192377. http://care.diabetesjournals.org/cgi/content/full/30/suppl_1/S4.
- ^ a b Eurich DT, McAlister FA, Blackburn DF, et al. (2007). "Benefits and harms of antidiabetic agents in patients with diabetes and heart failure: systematic review". BMJ 335 (7618): 497. doi:. PMID 17761999. http://www.bmj.com/cgi/content/full/335/7618/497.
- ^ Top 200 Generic Drugs by Units in 2006PDF (19.4 KB). Drug Topics (March 5, 2007). Retrieved on April 8, 2007.
- ^ (March 2007) WHO Model List of Essential MedicinesPDF (612 KB), 15th edition, World Health Organization, p. 21. Retrieved on 2007-11-19.
- ^ Witters L (2001). "The blooming of the French lilac". J Clin Invest 108 (8): 1105–7. doi:. PMID 11602616. Full text at PMC: 209536
- ^ Ungar G, Freedman L, Shapiro S (1957). "Pharmacological studies of a new oral hypoglycemic drug". Proc Soc Exp Biol Med 95 (1): 190–2. PMID 13432032.
- ^ U.S. Food and Drug Administration (December 30, 1994). FDA Approves New Diabetes Drug. Press release. http://www.fda.gov/bbs/topics/ANSWERS/ANS00627.html. Retrieved on 2007-01-06.
- ^ GLUCOPHAGE Label and Approval History. U.S. Food and Drug Administration. Retrieved on 2007-01-08. Data available for download on FDA website.
- ^ Selvin E, Bolen S, Yeh HC, et al (October 2008). "Cardiovascular outcomes in trials of oral diabetes medications: a systematic review". Arch Intern Med 168 (19): 2070–80. doi:. PMID 18955635.
- ^ "Effect of intensive blood-glucose control with metformin on complications in overweight patients with type 2 diabetes (UKPDS 34). UK Prospective Diabetes Study (UKPDS) Group". Lancet 352 (9131): 854–65. 1998. doi:. PMID 9742977.
- ^ Kilo C, Mezitis N, Jain R, Mersey J, McGill J, Raskin P (2003). "Starting patients with type 2 diabetes on insulin therapy using once-daily injections of biphasic insulin aspart 70/30, biphasic human insulin 70/30, or NPH insulin in combination with metformin". J Diabetes Complications 17 (6): 307–13. doi:. PMID 14583174.
- ^ DiPiro, Joseph T.; Talbert, Robert L.; Yee, Gary C.; Matzke, Gary R.; Wells, Barbara G.; Posey, L. Michael (2005). Pharmacotherapy: a pathophysiologic approach. New York: McGraw-Hill. ISBN 0071416137.
- ^ Stumvoll M, Nurjhan N, Perriello G, Dailey G, Gerich JE (1995). "Metabolic effects of metformin in non-insulin-dependent diabetes mellitus". N Engl J Med 333 (9): 550–4. doi:. PMID 7623903. http://content.nejm.org/cgi/content/abstract/333/9/550.
- ^ a b Bolen S, Feldman L, Vassy J, et al (2007). "Systematic review: comparative effectiveness and safety of oral medications for type 2 diabetes mellitus". Ann Intern Med 147 (6): 386–99. PMID 17638715.
- ^ Lord JM, Flight IHK, Norman RJ (2003). "Metformin in polycystic ovary syndrome: systematic review and meta-analysis". BMJ 327 (7421): 951–3. doi:. PMID 14576245. http://www.bmj.com/cgi/content/full/327/7421/951.
- ^ Marchesini G, Brizi M, Bianchi G, Tomassetti S, Zoli M, Melchionda N (2001). "Metformin in non-alcoholic steatohepatitis". Lancet 358 (9285): 893–4. doi:. PMID 11567710.
- ^ Ibáñez L, Ong K, Valls C, Marcos MV, Dunger DB, de Zegher F (2006). "Metformin treatment to prevent early puberty in girls with precocious pubarche". J. Clin. Endocrinol. Metab. 91 (8): 2888–91. doi:. PMID 16684823.
- ^ National Institute for Health and Clinical Excellence. 11 Clinical guideline 11 : Fertility: assessment and treatment for people with fertility problems . London, 2004.
- ^ Nair S, Diehl AM, Wiseman M, Farr GH Jr, Perrillo RP (2004). "Metformin in the treatment of non-alcoholic steatohepatitis: a pilot open label trial". Aliment Pharmacol Ther 20 (1): 23–28. doi:. PMID 15225167.
- ^ Wu RR, Zhao JP, Jin H, et al (2008). "Lifestyle intervention and metformin for treatment of antipsychotic-induced weight gain: a randomized controlled trial". JAMA 299 (2): 185–93. doi:. PMID 18182600.
- ^ Jones G, Macklin J, Alexander W (2003). "Contraindications to the use of metformin". BMJ 326 (7379): 4–5. doi:. PMID 12511434. http://www.bmj.com/cgi/content/full/326/7379/4.
- ^ a b Weir J (March 19, 1999). Guidelines with Regard to Metformin-Induced Lactic Acidosis and X-ray Contrast Medium Agents. Royal College of Radiologists. Retrieved on 2007-10-26 through the Internet Archive.
- ^ a b Thomsen HS, Morcos SK (2003). "Contrast media and the kidney: European Society of Urogenital Radiology (ESUR) guidelines". Br J Radiol 76 (908): 513–8. doi:. PMID 12893691. http://bjr.birjournals.org/cgi/content/full/76/908/513.
- ^ Salpeter S, Greyber E, Pasternak G, Salpeter E (2003). "Risk of fatal and nonfatal lactic acidosis with metformin use in type 2 diabetes mellitus: systematic review and meta-analysis". Arch Intern Med 163 (21): 2594–602. doi:. PMID 14638559.
- ^ Drug Facts and Comparisons 2005 (59th edition ed.). Lippincott Williams & Wilkins. October 2004. ISBN 1-57439-193-3.
- ^ Wulffele MG, Kooy A, Lehert P, Bets D, Ogterop JC, Borger van der Burg B, Donker AJ, Stehouwer CD. (November 2003). "Effects of short-term treatment with metformin on serum concentrations of homocysteine, folate and vitamin B12 in type 2 diabetes mellitus: a randomized, placebo-controlled trial". J Intern Med 254 (5): 455–63. doi:. PMID 14535967.
- ^ Andrès E, Noel E, Goichot B (2002). "Metformin-associated vitamin B12 deficiency". Arch Intern Med 162 (19): 2251–2. doi:. PMID 12390080.
- ^ Gilligan M (2002). "Metformin and vitamin B12 deficiency". Arch Intern Med 162 (4): 484–5. doi:. PMID 11863489.
- ^ Ting R, Szeto C, Chan M, Ma K, Chow K (2006). "Risk factors of vitamin B(12) deficiency in patients receiving metformin". Arch Intern Med 166 (18): 1975–9. doi:. PMID 17030830.
- ^ Spiller HA, Quadrani DA (2004). "Toxic effects from metformin exposure". The Annals of pharmacotherapy 38 (5): 776–80. doi:. PMID 15031415.
- ^ Gjedde S, Christiansen A, Pedersen SB, Rungby J (2003). "Survival following a metformin overdose of 63 g: a case report". Pharmacol. Toxicol. 93 (2): 98–9. doi:. PMID 12899672.
- ^ Harvey B, Hickman C, Hinson G, Ralph T, Mayer A (2005). "Severe lactic acidosis complicating metformin overdose successfully treated with high-volume venovenous hemofiltration and aggressive alkalinization". Pediatr Crit Care Med 6 (5): 598–601. PMID 16148825.
- ^ Guo PY, Storsley LJ, Finkle SN (2006). "Severe lactic acidosis treated with prolonged hemodialysis: recovery after massive overdoses of metformin". Semin Dial 19 (1): 80–3. doi:. PMID 16423187.
- ^ Kirpichnikov D, McFarlane SI, Sowers JR (2002). "Metformin: an update" (PDF). Ann Intern Med 137 (1): 25–33. PMID 12093242. http://www.annals.org/cgi/reprint/137/1/25.pdf. Retrieved on 2008-12-30.
- ^ Hundal R, Krssak M, Dufour S, Laurent D, Lebon V, Chandramouli V, Inzucchi S, Schumann W, Petersen K, Landau B, Shulman G (2000). "Mechanism by which metformin reduces glucose production in type 2 diabetes" (PDF). Diabetes 49 (12): 2063–9. doi:. PMID 11118008. http://diabetes.diabetesjournals.org/cgi/reprint/49/12/2063.
- ^ Towler MC, Hardie DG (2007). "AMP-activated protein kinase in metabolic control and insulin signaling". Circ Res 100 (3): 328–41. doi:. PMID 17307971. http://circres.ahajournals.org/cgi/content/full/100/3/328.
- ^ Zhou G, Myers R, Li Y, Chen Y, Shen X, Fenyk-Melody J, Wu M, Ventre J, Doebber T, Fujii N, Musi N, Hirshman M, Goodyear L, Moller D (2001). "Role of AMP-activated protein kinase in mechanism of metformin action". J Clin Invest 108 (8): 1167–74. doi:. PMID 11602624. http://www.jci.org/cgi/content/full/108/8/1167#SEC4.
- ^ Kim YD, Park KG, Lee YS, et al. (2008). "Metformin inhibits hepatic gluconeogenesis through AMP-activated protein kinase-dependent regulation of the orphan nuclear receptor SHP". Diabetes 57 (2): 306–14. doi:. PMID 17909097. http://diabetes.diabetesjournals.org/cgi/content/full/57/2/306.
- ^ Zhang L, He H, Balschi JA (2007). "Metformin and phenformin activate AMP-activated protein kinase in the heart by increasing cytosolic AMP concentration". Am J Physiol Heart Circ Physiol 293 (1): H457–66. doi:. PMID 17369473. http://ajpheart.physiology.org/cgi/content/full/293/1/H457.
- ^ Collier CA, Bruce CR, Smith AC, Lopaschuk G, Dyck DJ (2006). "Metformin counters the insulin-induced suppression of fatty acid oxidation and stimulation of triacylglycerol storage in rodent skeletal muscle". Am J Physiol Endocrinol Metab 291 (1): E182-E189. doi:. PMID 16478780.
- ^ Bailey CJ, Turner RC (1996). "Metformin". N Engl J Med 334 (9): 574–9. doi:. PMID 8569826.
- ^ Musi N, Hirshman MF, Nygren J, et al. (2002). "Metformin increases AMP-activated protein kinase activity in skeletal muscle of subjects with type 2 diabetes". Diabetes 51 (7): 2074–81. doi:. PMID 12086935. http://diabetes.diabetesjournals.org/cgi/content/full/51/7/2074.
- ^ Saeedi R, Parsons HL, Wambolt RB, et al. (2008). "Metabolic actions of metformin in the heart can occur by AMPK-independent mechanisms". Am J Physiol Heart Circ Physiol 294 (6): H2497–506. doi:. PMID 18375721.
- ^ Heller, Jacqueline B. (2007). "Metformin overdose in dogs and cats". Veterinary Medicine (April): 231–233. http://www.aspca.org/site/DocServer/vetm0407_231-234.pdf?docID=11061.
- ^ Somogyi A, Stockley C, Keal J, Rolan P, Bochner F (1987). "Reduction of metformin renal tubular secretion by cimetidine in man". Br J Clin Pharmacol 23 (5): 545–51. PMID 3593625.
- ^ a b "Glucophage Side Effects & Drug Interactions". RxList.com. 2007. http://www.rxlist.com/cgi/generic/metformi_ad.htm. Retrieved on 2007-11-19.
- ^ Jayasagar G, Krishna Kumar M, Chandrasekhar K, Madhusudan Rao C, Madhusudan Rao Y (2002). "Effect of cephalexin on the pharmacokinetics of metformin in healthy human volunteers". Drug Metabol Drug Interact 19 (1): 41–8. PMID 12222753.
- ^ GlaxoSmithKline (October 12, 2002). FDA Approves GlaxoSmithKline's Avandamet (rosiglitazone maleate and metformin HCl), The Latest Advancement in the Treatment of Type 2 Diabetes. Press release. http://www.docguide.com/news/content.nsf/news/8525697700573E1885256C4F0075B2B3. Retrieved on 2006-12-27.
- ^ U.S. Food and Drug Administration (March 4, 2005). Questions and Answers about the Seizure of Paxil CR and Avandamet. Press release. http://www.fda.gov/oc/qanda/PaxilandAvandamet.html. Retrieved on 2006-12-27.
- ^ Reuters (September 27, 2007). Teva Pharm announces settlement of generic Avandia, Avandamet, and Avandaryl litigation with GlaxoSmithKline. Press release. http://www.reuters.com/article/inPlayBriefing/idUSIN20070927170530TEVA20070927. Retrieved on 2009-02-17.
[edit] External links
- Metformin at the Open Directory Project
- Metformin drug information from Lexi-Comp. Includes dosage information and a comprehensive list of international brand names
|
|||||||||||||||||||||||||||||||||||||||||||||||||