Silicon
From Wikipedia, the free encyclopedia
|
|||||||||||||||||||||||||||||||
| General | |||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Name, symbol, number | silicon, Si, 14 | ||||||||||||||||||||||||||||||
| Element category | metalloid | ||||||||||||||||||||||||||||||
| Group, period, block | 14, 3, p | ||||||||||||||||||||||||||||||
| Appearance | crystalline, reflective | ||||||||||||||||||||||||||||||
| Standard atomic weight | 28.0855(3) g·mol−1 | ||||||||||||||||||||||||||||||
| Electron configuration | [Ne] 3s2 3p2 | ||||||||||||||||||||||||||||||
| Electrons per shell | 2, 8, 4 (Image) | ||||||||||||||||||||||||||||||
| Physical properties | |||||||||||||||||||||||||||||||
| Phase | solid | ||||||||||||||||||||||||||||||
| Density (near r.t.) | 2.3290 g·cm−3 | ||||||||||||||||||||||||||||||
| Liquid density at m.p. | 2.57 g·cm−3 | ||||||||||||||||||||||||||||||
| Melting point | 1687 K (1414 °C, 2577 °F) |
||||||||||||||||||||||||||||||
| Boiling point | 3538 K (3265 °C, 5909 °F) |
||||||||||||||||||||||||||||||
| Heat of fusion | 50.21 kJ·mol−1 | ||||||||||||||||||||||||||||||
| Heat of vaporization | 359 kJ·mol−1 | ||||||||||||||||||||||||||||||
| Specific heat capacity | (25 °C) 19.789 J·mol−1·K−1 | ||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||
| Atomic properties | |||||||||||||||||||||||||||||||
| Crystal structure | Diamond cubic | ||||||||||||||||||||||||||||||
| Oxidation states | 4, 3 [1], 2 [2], 1 [3] (amphoteric oxide) |
||||||||||||||||||||||||||||||
| Electronegativity | 1.90 (Pauling scale) | ||||||||||||||||||||||||||||||
| Ionization energies (more) |
1st: 786.5 kJ·mol−1 | ||||||||||||||||||||||||||||||
| 2nd: 1577.1 kJ·mol−1 | |||||||||||||||||||||||||||||||
| 3rd: 3231.6 kJ·mol−1 | |||||||||||||||||||||||||||||||
| Atomic radius | 117.6 pm | ||||||||||||||||||||||||||||||
| Atomic radius (calc.) | 111 pm | ||||||||||||||||||||||||||||||
| Covalent radius | 111 pm | ||||||||||||||||||||||||||||||
| Van der Waals radius | 210 pm | ||||||||||||||||||||||||||||||
| Miscellaneous | |||||||||||||||||||||||||||||||
| Magnetic ordering | nonmagnetic | ||||||||||||||||||||||||||||||
| Thermal conductivity | (300 K) 149 W·m−1·K−1 | ||||||||||||||||||||||||||||||
| Thermal expansion | (25 °C) 2.6 µm·m−1·K−1 | ||||||||||||||||||||||||||||||
| Speed of sound (thin rod) | (20 °C) 8433 m/s | ||||||||||||||||||||||||||||||
| Young's modulus | 150 GPa | ||||||||||||||||||||||||||||||
| Bulk modulus | 100 GPa | ||||||||||||||||||||||||||||||
| Mohs hardness | 7 | ||||||||||||||||||||||||||||||
| CAS registry number | 7440-21-3 | ||||||||||||||||||||||||||||||
| Band gap energy at 300 K | 1.12 eV | ||||||||||||||||||||||||||||||
| Most stable isotopes | |||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||
| References | |||||||||||||||||||||||||||||||
Silicon (pronounced /ˈsɪlɨkən/, Latin: silicium) is the most common metalloid. It is a chemical element, which has the symbol Si and atomic number 14. The atomic mass is 28.0855. A tetravalent metalloid, silicon is less reactive than its chemical analog carbon. As the eighth most common element in the universe by mass, silicon very rarely occurs as the pure free element in nature, but is more widely distributed in dusts, planetoids and planets as various forms of silicon dioxide (silica) or silicates. On Earth, silicon is the second most abundant element (after oxygen) in the crust,[1] making up 25.7% of the crust by mass.
Silicon has many industrial uses. Elemental silicon is the principal component of most semiconductor devices, most importantly integrated circuits or microchips. Silicon is widely used in semiconductors because it remains a semiconductor at higher temperatures than the semiconductor germanium and because its native oxide is easily grown in a furnace and forms a better semiconductor/dielectric interface than any other material.
In the form of silica and silicates, silicon forms useful glasses, cements, and ceramics. It is also a constituent of silicones, a class-name for various synthetic plastic substances made of silicon, oxygen, carbon and hydrogen, often confused with silicon itself.
Silicon is an essential element in biology, although only tiny traces of it appear to be required by animals[citation needed]. It is much more important to the metabolism of plants, particularly many grasses, and silicic acid (a type of silica) forms the basis of the striking array of protective shells of the microscopic diatoms.
Contents |
[edit] Notable characteristics
The outer electron orbitals (half filled subshell holding up to eight electrons) have the same structure as in carbon and the two elements are sometimes similar chemically. Even though it is a relatively inert element, silicon still reacts with halogens and dilute alkalis, but most acids (except for some hyper-reactive combinations of nitric acid and hydrofluoric acid) do not affect it. Having four bonding electrons however gives it, like carbon, many opportunities to combine with other elements or compounds under the right circumstances.
Both silicon and (in certain aspects) carbon are semiconductors, readily either donating or sharing their four outer electrons allowing many different forms of chemical bonding. Pure silicon has a negative temperature coefficient of resistance, since the number of free charge carriers increases with temperature. The electrical resistance of single crystal silicon significantly changes under the application of mechanical stress due to the piezoresistive effect.
In its crystalline form, pure silicon has a gray color and a metallic luster. It is similar to glass in that it is rather strong, very brittle, and prone to chipping.
[edit] History
| Please help improve this article or section by expanding it. Further information might be found on the talk page. (February 2009) |
Silicon was first identified by Antoine Lavoisier in 1787 (as a component of the Latin silex, silicis for flint, flints), and was later mistaken by Humphry Davy in 1800 for a compound. In 1811 Gay-Lussac and Thénard probably prepared impure amorphous silicon through the heating of potassium with silicon tetrafluoride. In 1824, Berzelius prepared amorphous silicon using approximately the same method as Lussac. Berzelius also purified the product by repeatedly washing it.[2]
[edit] Occurrence
Measured by mass, silicon makes up 25.7% of the Earth's crust and is the second most abundant element in the crust, after oxygen. Pure silicon crystals are very rarely found in nature; they can be found as inclusions with gold and in volcanic exhalations. Silicon is usually found in the form of silicon dioxide (also known as quartz), and other more complex silicate minerals.
Silica occurs in minerals consisting of (practically) pure silicon dioxide in different crystalline forms. Amethyst, agate, quartz, rock crystal, chalcedony, flint, jasper, and opal are some of the forms in which silicon dioxide appears. Biogenic silica occurs in the form of diatoms, radiolaria and siliceous sponges.
Silicon also occurs as silicate minerals (various minerals containing silicon, oxygen and one or another metal), for example the feldspar group. These minerals occur in clay, sand and various types of rock such as granite and sandstone. Feldspar, pyroxene, amphibole, and mica are a few of the many common silicate mineral groups.
Silicon is a principal component of many meteorites, and also is a component of obsidian and tektites, which are natural forms of glass.
See also Category:Silicate minerals
[edit] Isotopes
| Please help improve this article or section by expanding it. Further information might be found on the talk page. (February 2009) |
Silicon has numerous known isotopes, with mass numbers ranging from 22 to 44. 28Si (the most abundant isotope, at 92.23%), 29Si (4.67%), and 30Si (3.1%) are stable; 32Si is a radioactive isotope produced by cosmic ray spallation of argon. Its half-life has been determined to be approximately 170 years (0.21 MeV), and it decays by beta - emission to 32P (which has a 14.28 day half-life )[3] and then to 32S.
[edit] Compounds
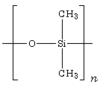
Silicon forms binary compounds called silicides with many metallic elements whose properties range from reactive compounds e.g. magnesium silicide , Mg2Si through to high melting refractory compounds such as molybdenum disilicide, MoSi2.[4] Silicon carbide, SiC, (carborundum) is a hard, high melting solid and is the well known abrasive. Silane, SiH4, is a pyrophoric gas with a similar tetrahedral structure to methane, CH4. Additionally there is a range of catenated silicon hydrides that form a homologous series of compounds, SinH2n+2 where n = 2-8 (analogous to the alkanes).[5] These are all readily hydrolysed and are thermally unstable, particularly the heavier members.[5] Disilenes contain a silicon-silicon double bond (analogous to the alkenes) and are generally highly reactive requiring large substituent groups to stabilise them.[6] A disilyne with a silicon-silicon triple bond was first isolated in 2004, although as the compound is non-linear, the bonding is dissimilar to that in alkynes.[7] Tetrahalides, SiX4, are formed with all of the halogens.[4] Silicon tetrachloride for example readily reacts with water unlike its carbon analogue, carbon tetrachloride.[5] Silicon dihalides are formed by the high temperature reaction of tetrahalides and silicon, with a structure analogous to a carbene they are reactive compounds. [5] Silicon difluoride condenses to form a polymeric compound, (SiF2)n.[5] Silicon dioxide, is a high melting solid with a number of different crystal forms the most familiar of which is the mineral quartz.[4] In quartz each silicon atom is surrounded by four oxygen atoms that bridge to other silicon atoms to form a three dimensional lattice.[4] Silica is soluble in water at high temperatures forming monosilicic acid (Si(OH)4)[5] and this property is used in the manufacture of quartz crystals used in electronics. ,[4]
Under the right conditions monosilicic acid readily polymerises to form more complex silicic acids, ranging from the simplest condensate, disilicic acid (H6Si2O7) to linear, ribbon, layer and lattice structures which form the basis of the many different silicate minerals.[5] Silicates are also important constituents of concretes.[4] With oxides of other elements the high temperature reaction of silicon dioxide can give a wide range of glasses with various properties.[5] Examples include soda lime glass, borosilicate glass and lead crystal glass. Silicon sulfide, SiS2 is a polymeric solid (unlike its carbon analogue the liquid CS2).[4] Silicon forms a nitride, Si3N4 which is a ceramic.[4] Silatranes, a group of tricyclic compounds containing five coordinate silicon may have physiological properties.[8] Many transition metal complexes containing a metal-silicon bond are now known, which include complexes containing, SiHnX3-n ligands, SiX3 ligands, Si(OR)3 ligands.[8] Silicones are large group of polymeric compounds with an (Si-O-Si) backbone an example is the silicone oil, PDMS, polydimethylsiloxane).[4] These polymers can be crosslinked to produce resins and elestomers.[4] Many organosilicon compounds are known which contain a silicon-carbon single bond. Many of these are based on a central tetrahedral silicon atom and some are optically active when central chirality exists. Long chain polymers containing a silicon backbone are known for example polydimethysilylene (SiMe2)n.[9] Polycarbosilane, [(SiMe2)2CH2]n with a backbone containing a repeating -Si-Si-C unit is a precursor in the production of silicon carbide fibres.[9]
[edit] Applications
As the second most abundant element in the earth's crust, silicon is vital to the construction industry as a principal constituent of natural stone, glass, concrete and cement. Silicon's greatest impact on the modern world's economy and lifestyle has resulted from silicon wafers used as substrates in the manufacture of discrete electronic devices such as power transistors, and in the development of integrated circuits such as computer chips.
[edit] Alloys
The largest application of pure silicon (metallurgical grade silicon), representing about 55% of the world consumption, is in the manufacture of aluminium-silicon alloys to produce cast parts, mainly for the automotive industry. Silicon is an important constituent of electrical steel, modifying its resistivity and ferromagnetic properties. Silicon is added to molten cast iron as ferrosilicon or silicocalcium alloys to improve its performance in casting thin sections, and to prevent the formation of cementite at the surface.
[edit] In electronic applications
Pure silicon is used to produce ultra-pure silicon wafers used in the semiconductor industry, in electronics and in photovoltaic applications. Ultra-pure silicon can be doped with other elements to adjust its electrical response by controlling the number and charge (positive or negative) of current carriers. Such control is necessary for transistors, solar cells, integrated circuits, microprocessors, semiconductor detectors and other semiconductor devices which are used in electronics and other high-tech applications. In Photonics, silicon can be used as a continuous wave Raman laser medium to produce coherent light, though it is ineffective as a light source. Hydrogenated amorphous silicon is used in the production of low-cost, large-area electronics in applications such as LCDs, and of large-area, low-cost thin-film solar cells.
[edit] Silicones
The second largest application of silicon (about 40% of world consumption) is as a raw material in the production of silicones, compounds containing silicon-oxygen and silicon-carbon bonds that have the capability to act as bonding intermediates between glass and organic compounds, and to form polymers with useful properties such as impermeability to water, flexibility and resistance to chemical attack. Silicones are used in waterproofing treatments, molding compounds and mold-release agents, mechanical seals, high temperature greases and waxes, caulking compounds and even in applications as diverse as breast implants, contact lenses, explosives and pyrotechnics.[10]
- Construction: Silicon dioxide or silica in the form of sand and clay is an important ingredient of concrete and brick and is also used to produce Portland cement.
- Pottery/Enamel is a refractory material used in high-temperature material production and its silicates are used in making enamels and pottery.
- Glass: Silica from sand is a principal component of glass. Glass can be made into a great variety of shapes and with many different physical properties. Silica is used as a base material to make window glass, containers, insulators, and many other useful objects.
- Abrasives: Silicon carbide is one of the most important abrasives.
- Silly Putty was originally made by adding boric acid to silicone oil. Now name-brand Silly Putty also contains significant amounts of elemental silicon. (Silicon binds to the silicone and allows the material to bounce 20% higher.)[citation needed]
See also Category:Silicon compounds
[edit] Production
Silicon is commercially prepared by the reaction of high-purity silica with wood, charcoal, and coal, in an electric arc furnace using carbon electrodes. At temperatures over 1,900 °C (3,450 °F), the carbon reduces the silica to silicon according to the chemical equation
- SiO2 + C → Si + CO2.
- SiO2 + 2C → Si + 2CO.
Liquid silicon collects in the bottom of the furnace, and is then drained and cooled. The silicon produced via this process is called metallurgical grade silicon and is at least 98% pure. Using this method, silicon carbide, SiC, can form. However, provided the amount of SiO2 is kept high, silicon carbide may be eliminated, as explained by this equation:
- 2 SiC + SiO2 → 3 Si + 2 CO.
In September 2008, metallurgical grade silicon cost about $1.45 per pound ($3.20/kg),[11] up from $0.77 per pound ($1.70/kg) in 2005.[12]
It has been reported in recent years that, by molten salt electrolysis, pure silicon can be directly extracted from solid silica and this new electrolysis method, known as the FFC Cambridge Process, has the potential to produce directly the solar grade silicon without any CO2 emission at much lower energy consumption.[13][14][15]
[edit] Purification
The use of silicon in semiconductor devices demands a much greater purity than afforded by metallurgical grade silicon. Historically, a number of methods have been used to produce high-purity silicon.
[edit] Physical methods
Early silicon purification techniques were based on the fact that if silicon is melted and re-solidified, the last parts of the mass to solidify contain most of the impurities. The earliest method of silicon purification, first described in 1919 and used on a limited basis to make radar components during World War II, involved crushing metallurgical grade silicon and then partially dissolving the silicon powder in an acid. When crushed, the silicon cracked so that the weaker impurity-rich regions were on the outside of the resulting grains of silicon. As a result, the impurity-rich silicon was the first to be dissolved when treated with acid, leaving behind a more pure product.
In zone melting, also called zone refining, the first silicon purification method to be widely used industrially, rods of metallurgical grade silicon are heated to melt at one end. Then, the heater is slowly moved down the length of the rod, keeping a small length of the rod molten as the silicon cools and re-solidifies behind it. Since most impurities tend to remain in the molten region rather than re-solidify, when the process is complete, most of the impurities in the rod will have been moved into the end that was the last to be melted. This end is then cut off and discarded, and the process repeated if a still higher purity is desired.
[edit] Chemical methods
Today, silicon is purified by converting it to a silicon compound that can be more easily purified by distillation than in its original state, and then converting that silicon compound back into pure silicon. Trichlorosilane is the silicon compound most commonly used as the intermediate, although silicon tetrachloride and silane are also used. When these gases are blown over silicon at high temperature, they decompose to high-purity silicon.
At one time, DuPont produced ultra-pure silicon by reacting silicon tetrachloride with high-purity zinc vapors at 950 °C, producing silicon according to the chemical equation
- SiCl4 + 2 Zn → Si + 2 ZnCl2.
However, this technique was plagued with practical problems (such as the zinc chloride byproduct solidifying and clogging lines) and was eventually abandoned in favor of the Siemens process.
In the Siemens process, high-purity silicon rods are exposed to trichlorosilane at 1150 °C. The trichlorosilane gas decomposes and deposits additional silicon onto the rods, enlarging them according to chemical reactions like
- 2 HSiCl3 → Si + 2 HCl + SiCl4.
Silicon produced from this and similar processes is called polycrystalline silicon. Polycrystalline silicon typically has impurity levels of less than 10−9.
In 2006 REC announced construction of a plant based on fluidized bed technology using silane.[16]
- 3SiCl4 + Si + 2H2 → 4HSiCl3
- 4HSiCl3 → 3SiCl4 + SiH4
- SiH4 → Si + 2H2
[edit] Crystallization
Silicon, like carbon and other group IV elements form face-centered diamond cubic crystal structure. Silicon, in particular, forms a face-centered cubic structure with a lattice spacing of 5.430710 Å (0.5430710 nm).[17]
The majority of silicon crystals grown for device production are produced by the Czochralski process, (CZ-Si) since it is the cheapest method available and it is capable of producing large size crystals. However, silicon single-crystals grown by the Czochralski method contain impurities since the crucible which contains the melt dissolves. For certain electronic devices, particularly those required for high power applications, silicon grown by the Czochralski method is not pure enough. For these applications, float-zone silicon (FZ-Si) can be used instead. It is worth mentioning though, in contrast with CZ-Si method in which the seed is dipped into the silicon melt and the growing crystal is pulled upward, the thin seed crystal in the FZ-Si method sustains the growing crystal as well as the polysilicon rod from the bottom. As a result, it is difficult to grow large size crystals using the float-zone method. Today, all the dislocation-free silicon crystals used in semiconductor industry with diameter 300 mm or larger are grown by the Czochralski method with purity level significantly improved.
[edit] Different forms of silicon
One can notice the color change in silicon nanopowder. This is caused by the quantum effects which occur in particles of nanometric dimensions. See also Potential well, Quantum dot, and Nanoparticle.
[edit] In popular culture
Because silicon is an important element in semiconductors and high-tech devices, the high-tech region of Silicon Valley, California is named after this element. Other geographic locations with connections to the industry have since characterized themselves as Siliconia as well, for example Silicon Saxony in Germany.
[edit] See also
- Black silicon
- Covalent superconductors
- Crystalline silicon
- Electronics
- List of silicon producers
- Printed silicon electronics
- RCA clean
- Silicone
- Wafer (electronics)
[edit] References
- ^ "The periodic table". webelements.com. http://www.webelements.com/. Retrieved on 2008-02-20.
- ^ Weeks, Mary Elvira (1932). "The discovery of the elements: XII. Other elements isolated with the aid of potassium and sodium: beryllium, boron, silicon, and aluminum". Journal of Chemical Education: 1386–1412.
- ^ "Phosphorus - 32". sciencegateway.org. http://sciencegateway.org/isotope/phosp32.html. Retrieved on 2008-02-20.
- ^ a b c d e f g h i j Greenwood, N. N.; Earnshaw, A. (1997). Chemistry of the Elements (2nd Edition ed.). Oxford:Butterworth-Heinemann. ISBN 0-7506-3365-4.
- ^ a b c d e f g h Holleman, A. F.; Wiberg, E.; Wiberg, N. (2001). Inorganic Chemistry, 1st Edition. Academic Press. ISBN 0123526515.
- ^ (Multiply Bonded Main Group Metals and Metalloids: Multiple Bonded Main Group Metals and Metalloids, F. G. Stone, Robert West, Academic Press, 1996, ISBN 0120311399)
- ^ A Stable Compound Containing a Silicon-Silicon Triple Bond, Akira Sekiguchi, Rei Kinjo, Masaaki Ichinohe, Science 17 September 2004:Vol. 305. no. 5691, pp. 1755 - 1757 doi:10.1126/science.1102209
- ^ a b Lickiss, Paul D. (1994). Encyclopedia of Inorganic Chemistry. John Wiley & Sons. ISBN 0471936200.
- ^ a b Sakurai, Hideki (1994). Encyclopedia of Inorganic Chemistry. John Wiley & Sons. ISBN 0471936200.
- ^ Koch, E.C.; Clement, D.. Special Materials in Pyrotechnics: VI. Silicon - An Old Fuel with New Perspectives. http://www3.interscience.wiley.com/cgi-bin/abstract/114279686/ABSTRACT.
- ^ "Metallurgical silicon could become a rare commodity – just how quickly that happens depends to a certain extent on the current financial crisis". Photon International. http://www.photon-magazine.com/news_archiv/details.aspx?cat=News_PI&sub=worldwide&pub=4&parent=1555. Retrieved on 2009-03-04.
- ^ "Silicon". usgs.gov. http://minerals.usgs.gov/minerals/pubs/commodity/silicon/silicmcs06.pdf. Retrieved on 2008-02-20.
- ^ "New silicon production method with no carbon reductant". http://www.acr.net.au/~coastwatchers/charcoalition/noreductant.html. Referering to Chen, George Zheng; Fray, Derek J.; Farthing, Tom W. (2000). "Direct electrochemical reduction of titanium dioxide to titanium in molten calcium chloride". Nature 407 (6802): 361–364. doi:.
- ^ Xianbo Jin, Pei Gao, Dihua Wang, Xiaohong Hu, George Z. Chen (2004). "Electrochemical Preparation of Silicon and Its Alloys from Solid Oxides in Molten Calcium Chloride". Angew. Chem. Int. Ed. 43: 733–736.
- ^ Nohira, Toshiyuki; Yasuda, Kouji; Ito, Yasuhiko (2003). "Pinpoint and bulk electrochemical reduction of insulating silicon dioxide to silicon". Nature Materials 2: 397–401. doi:.
- ^ "Analyst_silicon_field_trip_March_28,_2007". hugin.info. http://hugin.info/136555/R/1115224/203491.pdf. Retrieved on 2008-02-20.
- ^ O'Mara, William C. (1990). Handbook of Semiconductor Silicon Technology. William Andrew Inc.. p. 349-352. ISBN 0815512376. http://books.google.com/books?id=COcVgAtqeKkC&pg=PA351&dq=Czochralski+Silicon+Crystal+Face+Cubic&lr=&as_brr=3&sig=ht-dgSy1lzBMYC7IXPp9W5QBqYo. Retrieved on 2008-02-24.
[edit] External links
| The Wikibook [[Wikibooks:|]] has a page on the topic of |
| Wikimedia Commons has media related to: Silicon |
| Look up silicon in Wiktionary, the free dictionary. |
| Periodic table | |||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| H | He | ||||||||||||||||||||||||||||||||||||||||
| Li | Be | B | C | N | O | F | Ne | ||||||||||||||||||||||||||||||||||
| Na | Mg | Al | Si | P | S | Cl | Ar | ||||||||||||||||||||||||||||||||||
| K | Ca | Sc | Ti | V | Cr | Mn | Fe | Co | Ni | Cu | Zn | Ga | Ge | As | Se | Br | Kr | ||||||||||||||||||||||||
| Rb | Sr | Y | Zr | Nb | Mo | Tc | Ru | Rh | Pd | Ag | Cd | In | Sn | Sb | Te | I | Xe | ||||||||||||||||||||||||
| Cs | Ba | La | Ce | Pr | Nd | Pm | Sm | Eu | Gd | Tb | Dy | Ho | Er | Tm | Yb | Lu | Hf | Ta | W | Re | Os | Ir | Pt | Au | Hg | Tl | Pb | Bi | Po | At | Rn | ||||||||||
| Fr | Ra | Ac | Th | Pa | U | Np | Pu | Am | Cm | Bk | Cf | Es | Fm | Md | No | Lr | Rf | Db | Sg | Bh | Hs | Mt | Ds | Rg | Uub | Uut | Uuq | Uup | Uuh | Uus | Uuo | ||||||||||
|
|||||||||||||||||||||||||||||||||||||||||
|
|||||