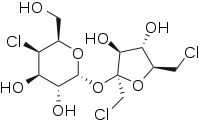
Sucralose
From Wikipedia, the free encyclopedia
| Sucralose[1] | |
|---|---|
 |
|
 |
|
| IUPAC name |
|
| Other names | 1',4,6'-Trichlorogalactosucrose Trichlorosucrose E955 |
| Identifiers | |
| CAS number | 56038-13-2 |
| EC number | |
| SMILES |
|
| ChemSpider ID | |
| Properties | |
| Molecular formula | C12H19Cl3O8 |
| Molar mass | 397.64 g/mol |
| Melting point |
130 °C |
| Except where noted otherwise, data are given for materials in their standard state (at 25 °C, 100 kPa) Infobox references |
|
Sucralose is a zero-calorie sugar substitute artificial sweetener. In the European Union, it is also known under the E number (additive code) E955. Sucralose is approximately 600 times as sweet as sucrose (table sugar),[2] twice as sweet as saccharin, and 3.3 times as sweet as aspartame. Unlike aspartame, it is stable under heat and over a broad range of pH conditions and can be used in baking or in products that require a longer shelf life. The commercial success of sucralose-based products stems from its favorable comparison to other low calorie sweeteners in terms of taste, stability, and safety.[3]
Sucralose is normally added by manufacturers in the very small quantities necessary. In the United States, a bulked-up product designed for domestic use and of similar appearance to granular sugar is sold under the names Splenda and SucraPlus.
Contents |
[edit] History
Sucralose was discovered in 1976 by scientists from Tate & Lyle, working with researchers Leslie Hough and Shashikant Phadnis at Queen Elizabeth College (now part of King's College London).[4] On a late-summer day, Phadnis was told to test the powder. Phadnis thought that Hough asked him to taste it, so he did.[4] He found the compound to be exceptionally sweet (the final formula was 600 times as sweet as sugar). They worked with Tate & Lyle for a year before settling down on the final formula.
Sucralose was first approved for use in Canada in 1991. Subsequent approvals came in Australia in 1993, in New Zealand in 1996, in the United States in 1998, and in the European Union in 2004. As of 2008, it had been approved in over 80 countries, including Mexico, Brazil, China, India and Japan.[5]
[edit] Production
Tate & Lyle manufactures sucralose at a plant in McIntosh, Alabama, with additional capacity under construction in Jurong, Singapore. It is manufactured by the selective chlorination of sucrose (table sugar), in which three of the hydroxyl groups are replaced with chlorine atoms to produce sucralose. In May 2008, Fusion Nutraceuticals launched a brand called SucraPlus to compete against Tate & Lyle's Splenda. Sucralose is now also manufactured in India using the same technology as described in Tate & Lyle's now-expired patents.
[edit] Product uses
Sucralose can be found in more than 4,500 food and beverage products. It is used because it is a no-calorie sweetener, does not promote dental caries,[6] and is safe for consumption by diabetics.[7] Sucralose is used as a replacement for, or in combination with, other artificial or natural sweeteners such as aspartame, acesulfame potassium or high-fructose corn syrup. Sucralose is used in products such as candy, breakfast bars and soft drinks. It is also used in canned fruits wherein water and sucralose take the place of much higher calorie corn syrup based additives. Sucralose mixed with maltodextrin or dextrose (both made from corn) as bulking agents is sold internationally by McNeil Nutritionals under the Splenda brand name. In the United States and Canada, this blend is increasingly found in restaurants, including McDonald's, Tim Hortons and Starbucks, in yellow packets, in contrast to the blue packets commonly used by aspartame and the pink packets used by those containing saccharin sweeteners; though in Canada yellow packets are also associated with the SugarTwin brand of cyclamate sweetener.
[edit] Cooking
Sucralose is a highly heat-stable artificial sweetener, allowing it to be used in many recipes with little or no sugar. Sucralose is available in a granulated form that allows for same-volume substitution with sugar. This mix of granulated sucralose includes fillers, all of which rapidly dissolve in liquids.[citation needed] Unlike sucrose which dissolves to a clear state, sucralose suspension in clear liquids such as water results in a cloudy state. For example, gelatin and fruit preserves made with sucrose have a satiny, near jewel-like appearance, whereas the same products made with sucralose (whether cooked or not) appear translucent and marginally glistening.[citation needed] While the granulated sucralose provides apparent volume-for-volume sweetness, the texture in baked products may be noticeably different. Sucralose is non-hygroscopic, meaning it does not attract moisture, which can lead to baked goods that are noticeably drier and manifesting a less dense texture than baked products made with sucrose. Unlike sucrose which melts when baked at high temperatures, sucralose maintains its granular structure when subjected to dry, high heat (e.g., in a 350 °F (177 °C) oven). Thus, in some baking recipes, such as crème brûlée, which require sugar sprinkled on top to partially or fully melt and crystallize, substituting sucralose will not result in the same surface texture, crispness, or crystalline structure.
[edit] Packaging and storage
Pure sucralose is sold in bulk, but not in quantities suitable for individual use, although some highly concentrated sucralose-water blends are available online, using 1/4 tsp per 1 cup of sweetness or roughly 1 part sucralose to 2 parts water.[8] Pure dry sucralose undergoes some decomposition at elevated temperatures. When it is in solution or blended with maltodextrin, it is slightly more stable.
Most products that contain sucralose add fillers and additional sweetener to bring the product to the approximate volume and texture of an equivalent amount of sugar. This is because sucralose is nearly 600 times as sweet as sucrose (table sugar).
[edit] Health and safety regulation
Sucralose has been accepted by several national and international food safety regulatory bodies, including the U.S. Food and Drug Administration (FDA), Joint Food and Agriculture Organization/World Health Organization Expert Committee on Food Additives, The European Union's Scientific Committee on Food, Health Protection Branch of Health and Welfare Canada and Food Standards Australia-New Zealand (FSANZ). Sucralose is the only artificial sweetener ranked as "safe" by the consumer advocacy group Center for Science in the Public Interest.[9][10] According to the Canadian Diabetes Association, one can consume 15 mg/kg/day of Sucralose "on a daily basis over a ... lifetime without any adverse effects".[11]
“In determining the safety of sucralose, the FDA reviewed data from more than 110 studies in humans and animals. Many of the studies were designed to identify possible toxic effects including carcinogenic, reproductive and neurological effects. No such effects were found, and FDA's approval is based on the finding that sucralose is safe for human consumption.”[12] For example, McNeil Nutritional LLC studies submitted as part of its U.S. FDA Food Additive Petition 7A3987, indicated that "in the 2-year rodent bioassays...there was no evidence of carcinogenic activity for either sucralose or its hydrolysis products...."[13]
[edit] Public health and safety concerns
Results from over 100 animal and clinical studies in the FDA approval process unanimously indicated a lack of risk associated with sucralose intake.[4] However, some adverse effects were seen at doses that significantly exceeded the estimated daily intake (EDI), which is 1.1 mg/kg/day.[14] When the EDI is compared to the intake at which adverse effects are seen, known as the highest no adverse effects limit (HNEL), at 1500 mg/kg/day,[14] there is a large margin of safety. The bulk of sucralose ingested does not leave the gastrointestinal tract and is directly excreted in the feces while 11-27% of it is absorbed.[2] The amount that is absorbed from the GI tract is largely removed from the blood stream by the kidneys and eliminated in the urine with 20-30% of the absorbed sucralose being metabolized.[2]
[edit] Thymus
Some concern has been raised about the effect of sucralose on the thymus. A report from the Australian National Industrial Chemicals Notification and Assessment Scheme (NICNAS) cites two studies on rats, both of which found "a significant decrease in mean thymus weight" at high doses.[15] The sucralose dose which caused the effects was 3000 mg/kg/day for 28 days. For a 150 lb (68.2 kg) human, this would mean an intake of nearly 205 grams of sucralose a day, which is equivalent to more than 17,200 individual Splenda packets/day for approximately one month. The dose required to provoke any immunological response was 750 mg/kg/day,[16] or 51 grams of sucralose per day, which is nearly 4,300 Splenda packets/day. After evaluation of this data and other toxicological fidings, the NICNAS report concludes that sucralose does not pose a hazard to public health.[15]
[edit] Environmental effects
According to one study, sucralose is digestible by a number of microorganisms and is broken down once released into the environment.[17] However, measurements by the Swedish Environmental Research Institute have shown that wastewater treatment has little effect on sucralose, which is present in wastewater effluents at levels of several μg/L.[18] There are no known eco-toxicological effects at such levels, but the Swedish Environmental Protection Agency warns that there may be a continuous increase in levels if the compound is only slowly degraded in nature.[19]
[edit] Organochlorides
Sucralose is a organochloride (or chlorocarbon), some of which are known to have significant toxicity to plants or animals, including humans[20]. Although some chlorocarbons are toxic, sucralose is not known to be toxic in small quantities and is extremely insoluble in fat; it cannot accumulate in fat like chlorinated hydrocarbons. In addition, sucralose does not break down or dechlorinate.[21] In addition, a variety of a simple chlorinated hydrocarbons including dichloromethane, chloroform, and carbon tetrachloride have been isolated from marine algae. [22][23][24]
[edit] Effects on P-glycoprotein
One study found evidence that high doses of Splenda (up to 1000 mg/kg) reduced the amount of good bacteria in the intestines of rats by up to 50%.[25] It also increased the pH level in the intestines, contributed to an increase in body weight and affected the levels of P-glycoprotein in the body in such a way that crucial health-related drugs could be rejected. The P-gp effect "could result in crucial medications used in chemotherapy for cancer patients, AIDS treatment and drugs for heart conditions being shunted back into the intestines rather than being absorbed by the body as intended."[26]
[edit] Other potential effects
One report suggests sucralose is a possible trigger for some migraine patients.[27] Another study published in the Journal of Mutation Research linked doses of sucralose equivalent to 11,450 packets per day in a person to DNA damage in mice.[28]
Allergic reactions to sucralose have not been documented, but individuals sensitive to either maltodextrin or dextrose should consult a physician about using any sweeteners containing these fillers.
[edit] Natural alternatives
Critics of sucralose often favor natural alternatives, including xylitol, maltitol, thaumatin, isomalt, stevia and siraitia. However, some natural substances are alleged to cause other potential problems,[29][30][31] and natural products generally do not undergo controlled trials before being allowed in food.[32]
[edit] See also
[edit] References
- ^ Merck Index, 11th Edition, 8854.
- ^ a b c Michael A. Friedman, Lead Deputy Commissioner for the FDA, Food Additives Permitted for Direct Addition to Food for Human Consumption; Sucralose Federal Register: 21 CFR Part 172, Docket No. 87F-0086, April 3, 1998
- ^ A Report on Sucralose from the Food Sanitation Council, The Japan Food Chemical Research Foundation
- ^ a b c Sucralose: An Overview, by Genevieve Frank, Penn State University
- ^ Splenda No Calorie Sweetener Fact Sheet, splenda.com
- ^ Food and Drug Administration (2006). "Food labeling: health claims; dietary noncariogenic carbohydrate sweeteners and dental caries.". Federal Register 71 (60): 15559–15564.
- ^ Facts About Sucralose, American Dietetic Association, 2006.
- ^ Sweetzfree is a clear liquid syrup base, highly concentrated, and made from 100% pure Sucralose in a purified water concentrate.
- ^ Nutrition Action Health Letter, Center for Science in the Public Interest, May 2008
- ^ Which additives are safe? Which aren't?, Center for Science in the Public Interest
- ^ Diabetes.ca Acceptable daily intake of sweeteners
- ^ FDA Talk Paper T98-16
- ^ FDA Final Rule, Food Additives Permitted for Direct Addition to Food for Human Consumption; Sucralose
- ^ a b Baird, I. M., Shephard, N. W., Merritt, R. J., & Hildick-Smith, G. (2000). "Repeated dose study of sucralose tolerance in human subjects". Food Chemical Toxicology 38 (Supplement 2): S123–S129.. doi:.
- ^ a b Report from NICNAS, The Australian Government regulator of industrial chemicals (PDF document)
- ^ USFDA Department of Health and Human Services, 1998
- ^ Labare, Michael P.; Alexander, Martin (1993). "Biodegradation of sucralose in samples of natural environments". Environmental Toxicology and Chemistry 12 (5): 797–804. doi:.
- ^ Measurements of Sucralose in the Swedish Screening Program 2007, Part I; Sucralose in surface waters and STP samples
- ^ Sötningsmedel sprids till miljön - Naturvårdsverket
- ^ [www.epa.gov/pesticides/safety/healthcare/handbook/Chap06.pdf]
- ^ Daniel JW, Renwick AG, Roberts A, Sims J. The metabolic fate of sucralose in rats. Food Chem Tox. 2000;38(S2): S115-S121.
- ^ Natural organochlorines in living organisms: chlorinated compounds, biosynthesized
- ^ Gordon W. Gribble (1998). "Naturally Occurring Organohalogen Compounds". Acc. Chem. Res. 31 (3): 141–152. doi:.
- ^ Gordon W. Gribble (1999). "The diversity of naturally occurring organobromine compounds". Chemical Society Reviews 28 (5): 335. doi:.
- ^ Abou-Donia MB, El-Masry EM, Abdel-Rahman AA, McLendon RE, Schiffman SS (2008). "Splenda alters gut microflora and increases intestinal p-glycoprotein and cytochrome p-450 in male rats". J. Toxicol. Environ. Health Part A 71 (21): 1415–29. doi:. PMID 18800291.
- ^ Citizens for Health press release
- ^ Patel, Rajendrakumar M. (September 2006). "Popular Sweetner Sucralose as a Migraine Trigger". Journal of Head and Face Pain 46: 1303. doi:. http://www.blackwell-synergy.com/doi/abs/10.1111/j.1526-4610.2006.00543_1.x.
- ^ Journal of Mutation Research - August 2002
- ^ Lynch BS, Tischler AS, Capen C, Munro IC, McGirr LM, McClain RM (1996). "Low digestible carbohydrates (polyols and lactose): significance of adrenal medullary proliferative lesions in the rat". Regul. Toxicol. Pharmacol. 23 (3): 256–97. doi:. PMID 8812969.
- ^ Nunes AP, Ferreira-Machado SC, Nunes RM, Dantas FJ, De Mattos JC, Caldeira-de-Araújo A (2007). "Analysis of genotoxic potentiality of stevioside by comet assay". Food Chem. Toxicol. 45 (4): 662–6. doi:.
- ^ Canimoğlu S, Rencüzoğullari E (2006). "The cytogenetic effects of food sweetener maltitol in human peripheral lymphocytes". Drug Chem Toxicol 29 (3): 269–78. doi:.
- ^ Dietary Supplement Health and Education Act of 1994, Public Law 103-417, 103rd Congress
[edit] External links
[edit] Science
|
|||||



